CHEM 1031 Chapter Notes - Chapter 3: Sulfide, Bromine, Oxyanion
Document Summary
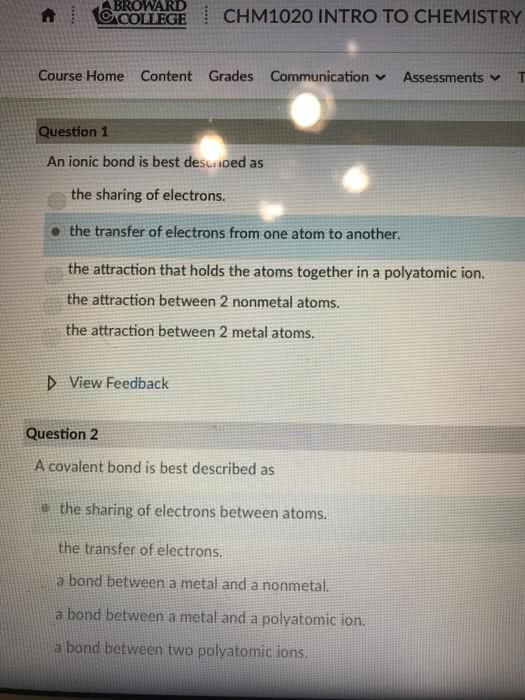
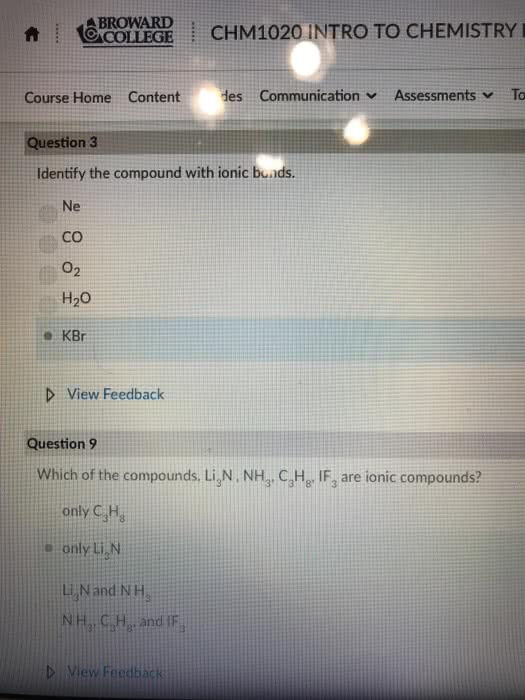
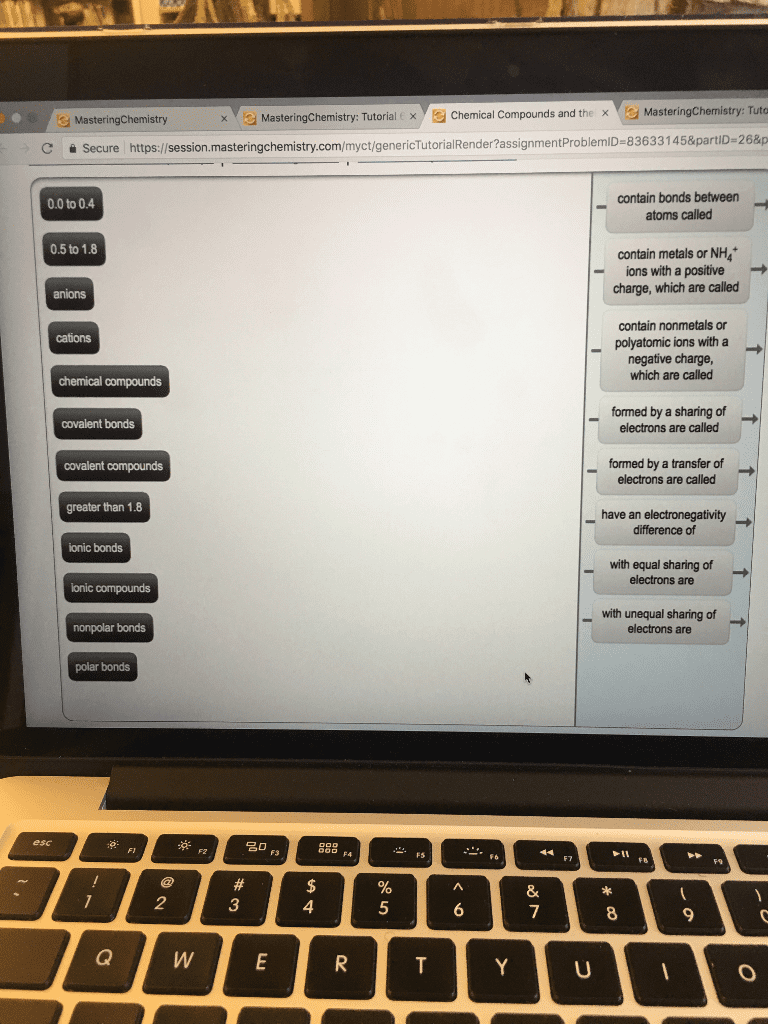
Compounds are composed of atoms held together by chemical bonds. Chemical bonds are the result of interaction between the charged particles electrons and protons that compose atoms. There are 2 types of bonds we will focus on ionic and covalent. Ionic bonds- occur between metals and nonmetals involving the transfer of electrons from one atom to the other. Covalent bonds- occur between two or more nonmetals involving the sharing of electrons between 2 atoms. Metals have a tendency to lose electrons and that nonmetal have a tendency to gain them. When a metal interacts with a nonmetal it can transfer one or more of its electrons to the nonmetal. The metal becomes a cation which is positively charged and the nonmetal becomes an anion a negatively charged ion. These oppositely charged ions are then attracted to one another by electrostatic forces and form and ionic bond. When a nonmetal bonds with another nonmetal, the atoms are sharing their electrons.