CHEM 1128Q Chapter Notes - Chapter 15: Boron, Manganese, Nonmetal

19
CHEM 1128Q Full Course Notes
Verified Note
19 documents
Document Summary
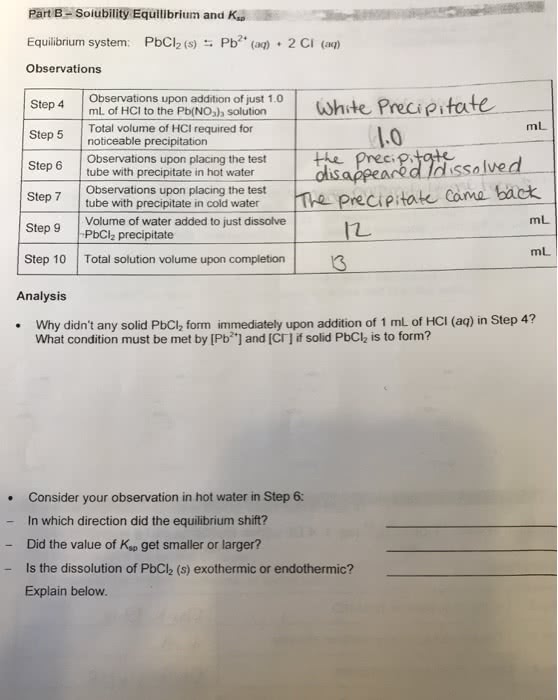
The dissolution of a slightly soluble ionic compound can be controlled by using le. Using an equilibrium constant can determine the concentration of ions present in solution. Ksp is the equilibrium constant between a slightly soluble ionic solid and a solution of its ions. The solid does not appear in the equilibrium expression for ksp. A small ksp value represents a system where the equilibrium lies to the left, so relatively few hydrated ions would be present in a saturated solution. A larger ksp means that the products are more and more favored, so the solid is more soluble. Since solubility is a measure of the maximum possible concentration of solute in a solution at a given t and p, ksp can be determined. Molar solubility is the solid"s solubility in m, moles/liter. Ksp can be derived from equilibrium concentrations. Solubility has to be converted into moles/liter (m) in order to use ksp.