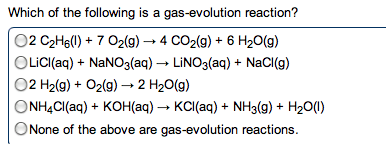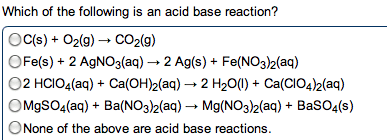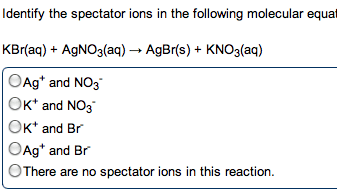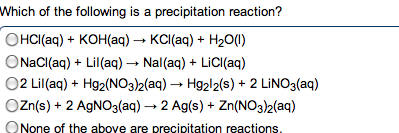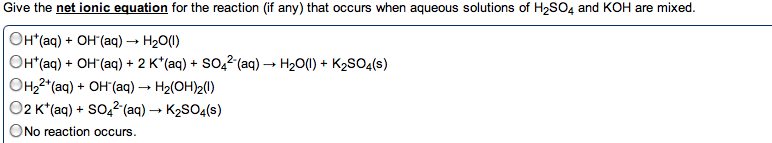CHEM 1AL Chapter Notes - Chapter 4: Erlenmeyer Flask, Alkali Metal, Chemical Equation
37 views1 pages
9 Nov 2018
School
Department
Course
Professor
Document Summary
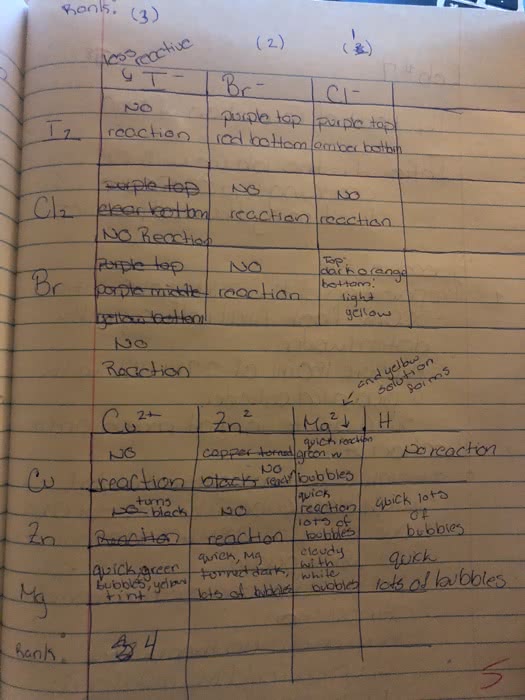
Metals: malleable, ductile, luster, high electrical and thermal conductivity. Tend to lose electrons to form cations. Alkali metals (group 1) and alkaline earth metals (group 2) are very reactive with non-metals (oxidation numbers of each of the reactants) Taken out for the net ionic equation because they cancel". Ions that appear on both sides of the total ionic reaction. Simplest way to show which species take part in a reaction. Metallic properties increase down a group and decrease across the row from left to right. Non-metals: tend to gain electrons to form anions. Base + acid -> salt + water. A substance that, when in solution, increases the concentration of oh - A substance that, when in solution, increases the concentration of h + Titration used to determine the concentration of an acid or base. Solution of known concentration is added to the unknown solution until the.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232