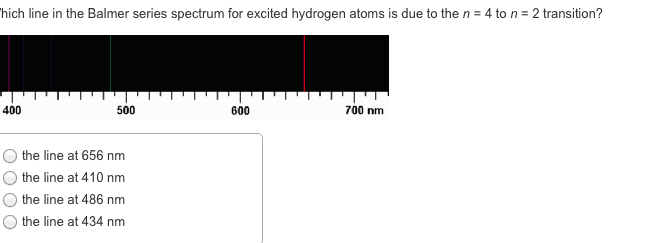
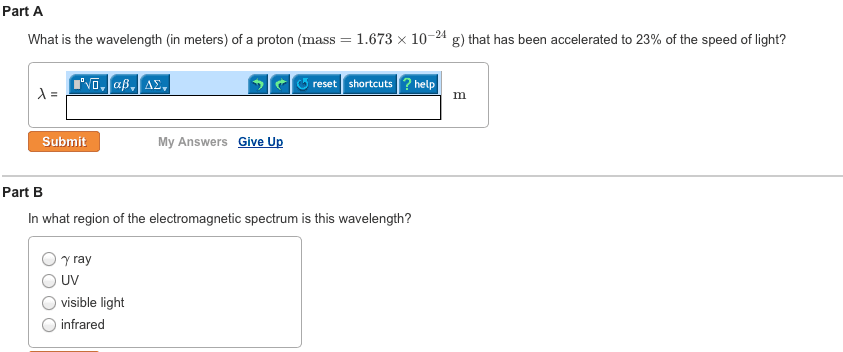
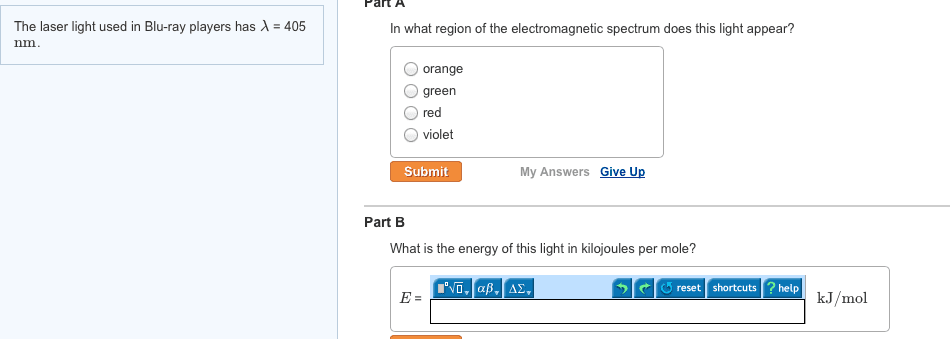
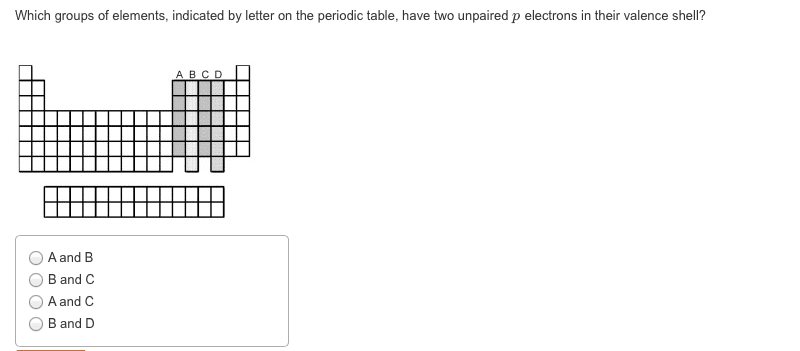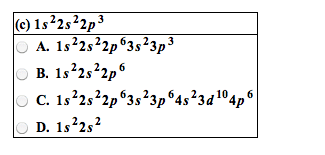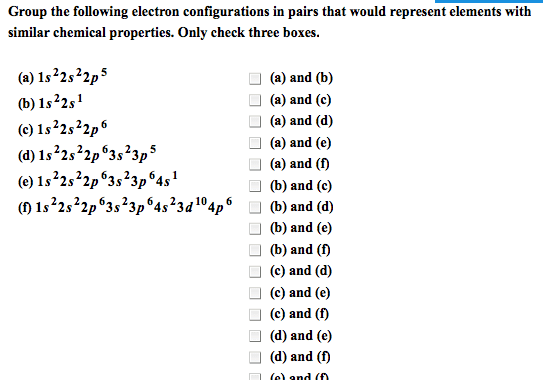CHEM 1061 Chapter Notes - Chapter 8: Ionic Radius, Ionic Compound, Core Electron
Document Summary
8. 2 the quantum-mechanical model and the periodic table. The lowest energy distribution of electrons in the sublevels of atoms. Recurring pattern in electron configurations, which is the basis for recurring patterns in chemical behaviors. Aufbau principle continue to add electrons to the lowest energy sublevel available. The orbital diagram: consists of a box for each orbital in a given energy level. Elements in the same group have similar outer electron configurations and similar patterns of reactivity. Sublevels are filled in order of increasing energy, which leads to outer electron configurations that recur periodically, which leads to chemical properties that recur periodically. Effects of shielding and penetrations: 3d orbital is filled in period 4, but the 4s orbital is filled first, 2s22p63s23p64s23d10. Inner (core) electrons: electrons that an atom has in common with the previous core electron. Outer electrons: those in the highest energy level, farthest from the nucleus. For main group elements, outer electrons = valence electrons.