CHEM 1110 Chapter Notes - Chapter 15: Reaction Quotient, Exothermic Process, Ideal Solution
Document Summary
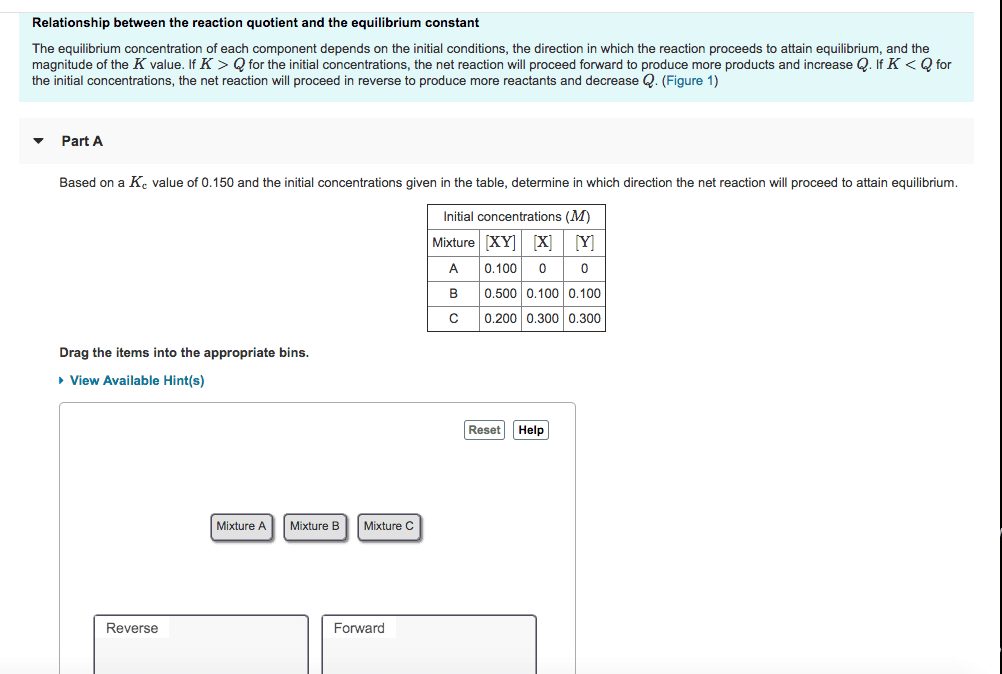
Chem 1110: principles of chemistry 1 chapter 15 reading notes. Chemical equilibrium occurs when opposing reactions proceed at equal rates. Just because the composition of the equilibrium mixture remains constant doesn"t mean they stop reacting it"s dynamic. At equilibrium, the concentration of reactions and products no longer change with time. For equilibrium to occur, nothing can escape from the system. Activity of a substance in the ideal mixture is the ratio of the concentration (m)/ pressure (atm) If k >> 1 (large k): equilibrium lies to the right, products predominate. If k<<1 (small k): equilibrium lies to the left, reactants predominate. It is forward and reverse reaction rates, not reactant and product concentrations that are equal at equilibrium. The equilibrium constant expression written in one direction is the reciprocal of the expression for the reaction written in is the reciprocal of the expression for the reaction written in the reverse direction.