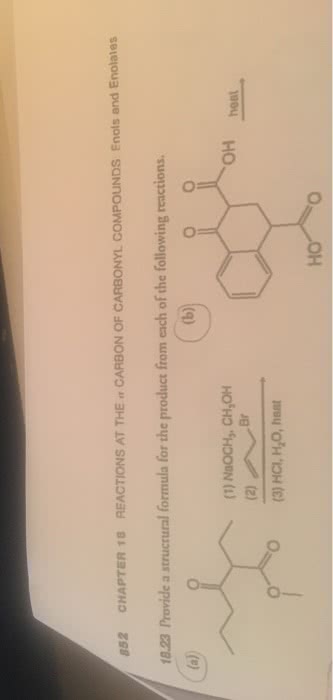
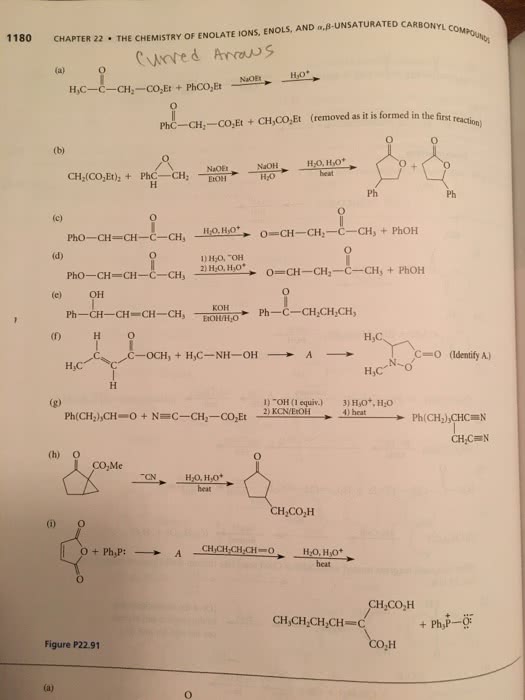
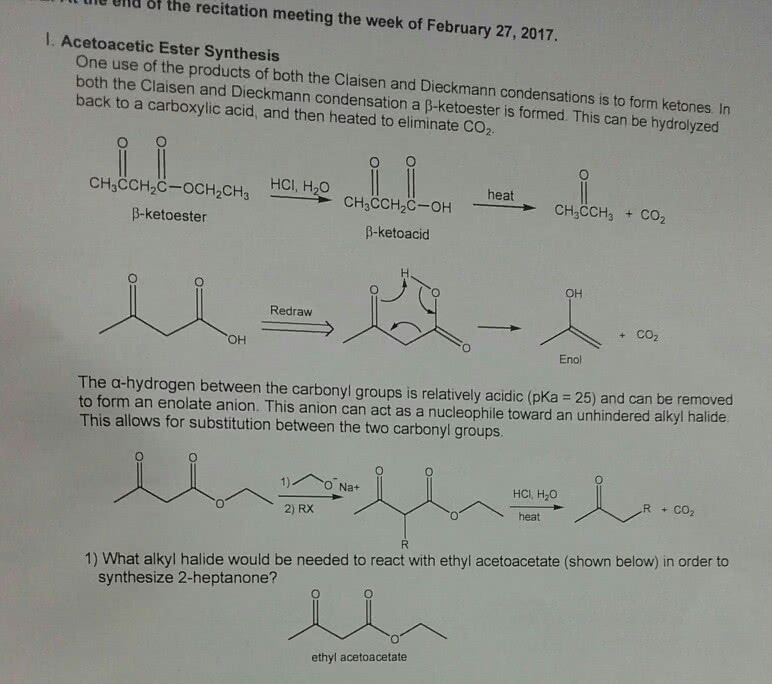
CHEM 242 Chapter 22: Chapter 22
Document Summary
Chapter 22 condensations and alpha substitutions of carbonyl compounds. Sometimes this equilibrium mixture of enolate a(cid:374)d (cid:271)ase wo(cid:374)"t work, usuall(cid:455) (cid:271)e(cid:272)ause the (cid:271)ase (h(cid:455)dro(cid:454)ide or alkoxide) reacts with the electrophile faster than the enolate does. In these cases, we need a base that reacts completely to convert the carbonyl compound to its enolate before adding the electrophile. The most effective and useful base for this purpose is lithium diisopropylamide (lda), the lithium salt of diisopropylamine: Enamines can displace halides from reactive alkyl halides, giving alkylated iminium salts. The iminium ions are unreactive toward further alkylation or acylation: In contrast with basic halogenation, acidic halogenation can selectively replace just one hydrogen or more than one, depending on the amount of the halogen added. Unlike ketones, aldehydes are easily oxidized, and halogens are strong oxidizing agents. Attempted halogenation of aldehydes usually results in oxidation to carboxylic acids: