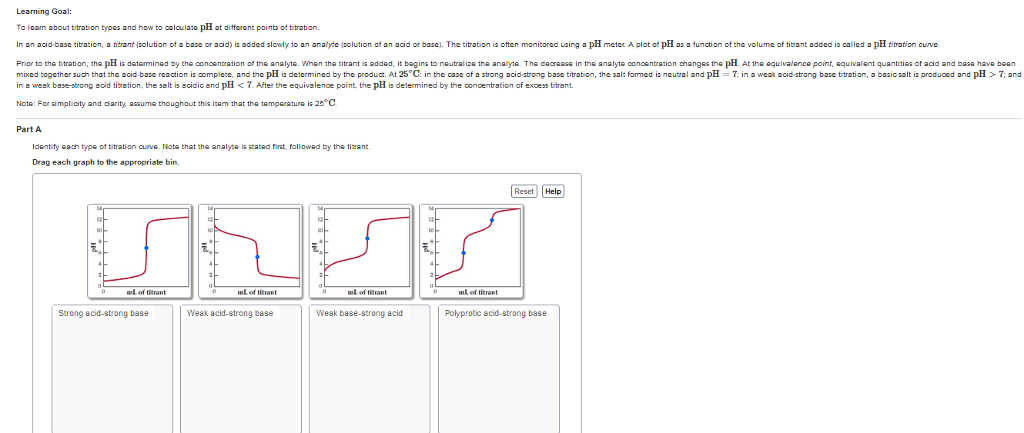
A lab experiment was conducted utilizing software to general a titration curve. 25 mL of an unknown analyte was titrated with .1015 M of NaOH solution. 
The pH at the equivalence point for solution 1 = 7.83.
12.61mL of NaOH titrant added.
The pH at the equivalence point for solution 2 = 8.53. Total volume of NaOH titrants for solution 1
12.47mL of NaOH titrant added.
What is Ka for the weak acid?
Please help with any calculations here.
Part 2 consists of unknown acid solutions with added base in which the pH was measured. Please help calculate the [A-]/[HA] of buffers.
Molar concentration of NaOH solution Solution 2 Volume of acid sample 12 mL Total volume of NaOH titrant l.) Moles of acid in sample: of acid: identity of acid (strong or weak) Calculated K, of weak acid Partil: Unknown Acid Solutions Volume of buffer sample: 13 Initial pH 62. 52 23 pH after mu addition of base: 413 pH after second tml. addition of base: IA of buffers: Note: The buffer solutions used in lab are an acetic acid/sodium acetate solution. The pK. of acetic acid is 4,75 on additional paper, clearly show an example of each calculation performed to fill in the report sheet. calculations are done in your lab notebook, photocopies are acceptable. Include sketches of all titration curves with correctly labeled axes, the correct coordinates for the equivalence point, where pH pK and correct volumes/pH values.