An abbreviated mechanistic description for DCC-mediated amide bond formation is illustrated as Path A. An alternative pathway for this process has been proposed as well, illustrated in Path B. In this alternative pathway, a second molecule of acid 1 out-competes the amine 4 for the pivotal activated acyl derivative 3, leading to a transient symmetrical anhydride 7 (with discharge of 6). In Path B, anhydride 7 then is the active electrophile that reacts with amine 4 to give the observed amide product 5 with regeneration of one molecule of acid 1.
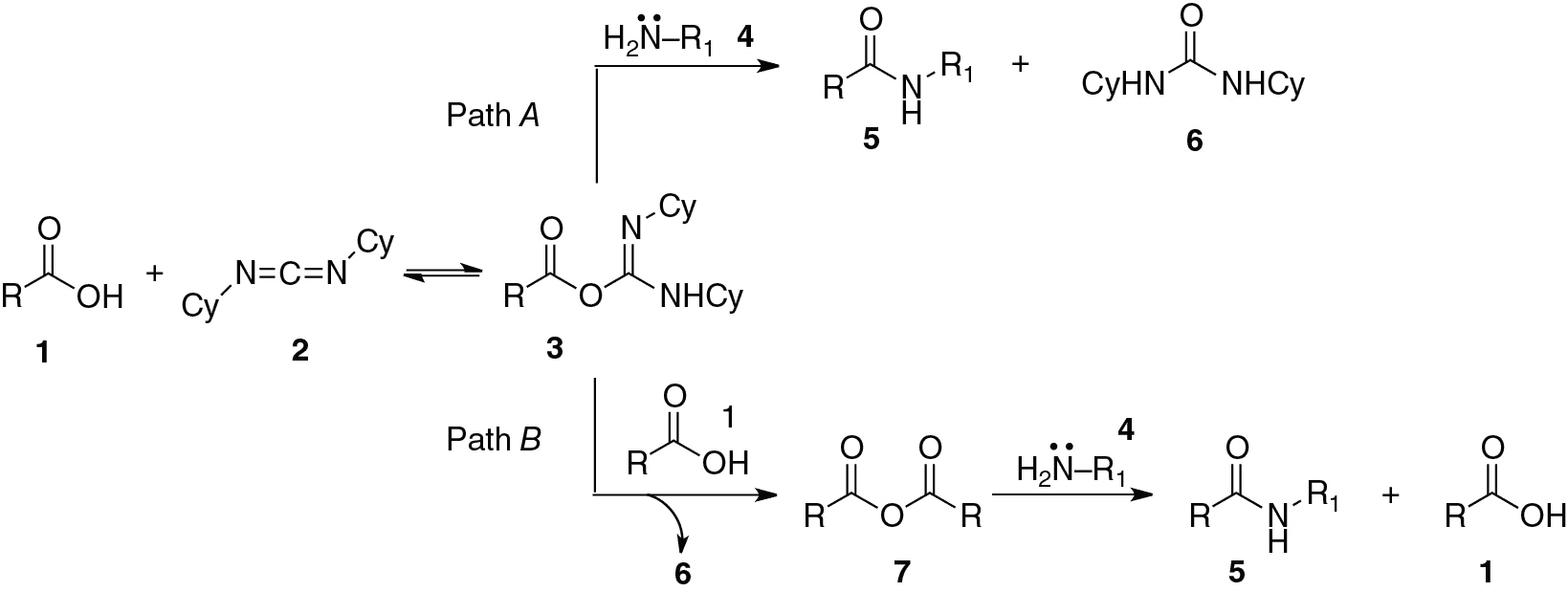
Assuming that only Path A were operational, what amide product(s) would you anticipate if the reaction were run with 1 equiv. of the achiral acid 8, 1 equiv. of the racemic amine 9, and a limiting amount (0.1 equiv.) of the chiral diimide 10?
O HM-R, HsN_R, R, + CyHN CyHN︿ NHCy Path A O ON Path B | R" \OH OO O O 7 R 4




