
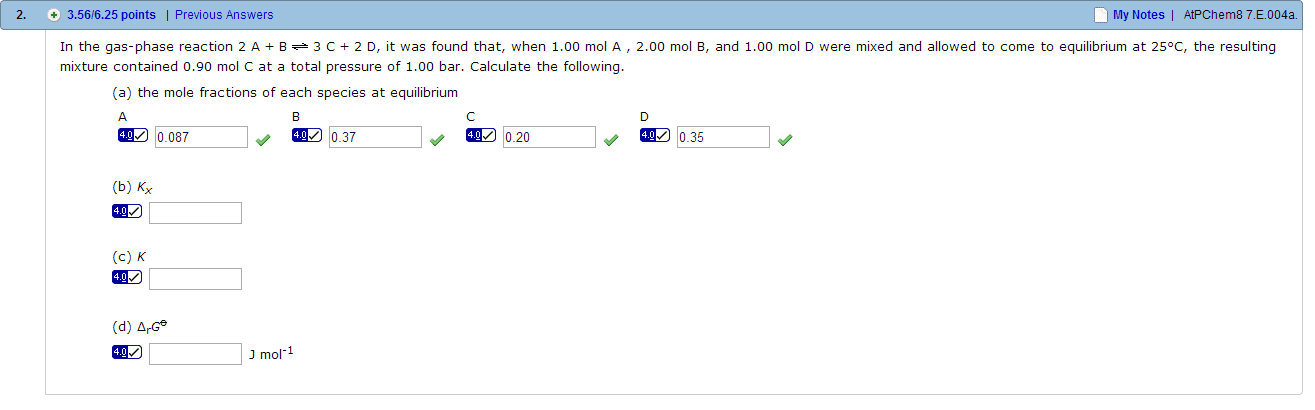
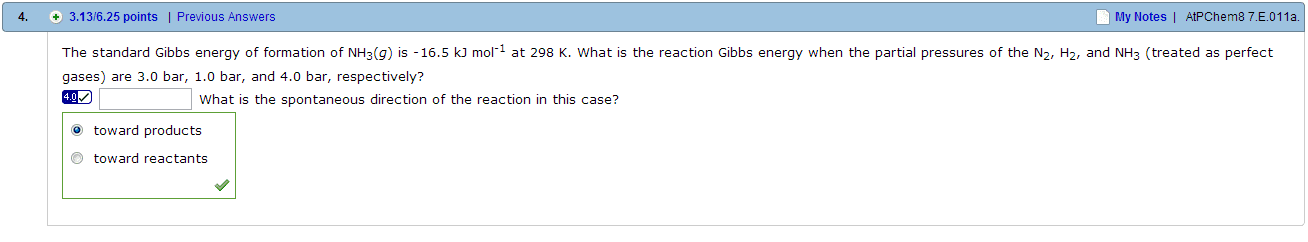
At 2257 K and 1.00 atm total pressure, water is 1.77 per cent dissociated at equilibrium by way of the reaction 2 H2O(g) 2H2(g) + O2(g). Calculate the following. K Delta r G0 kJ mol-1 Delta r G at this temperature kJ mol-1 In the gas-phase reaction 2A + B 3C + 2D, it was found that, when 1.00 mol A , 2.00 mol B, and 1.00 mol D were mixed and allowed to come to equilibrium at 25 degree C, the resulting mixture contained 0.90 mol C at a total pressure of 1.00 bar. Calculate the following. the mole fractions of each species at equilibrium A B D D Kx K Delta rG* J mol-1 The standard Gibbs energy of formation of NH3(g) is -16.5 kJ mol-1 at 298 K. What is the reaction Gibbs energy when the partial pressures of the N2, H2, and NH3 (treated as perfect gases) are 3.0 bar, 1.0 bar, and 4.0 bar, respectively? What is the spontaneous direction of the reaction in this case? toward products toward reactants





