CHEM 11200 Lecture Notes - Lecture 17: Heat Capacity
Document Summary
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
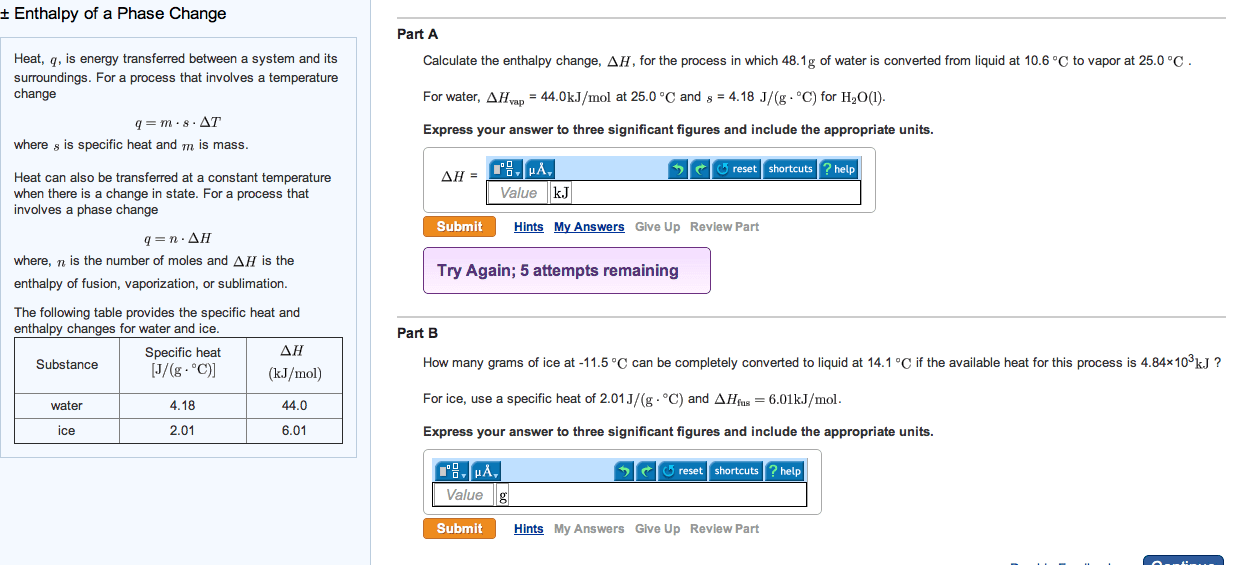
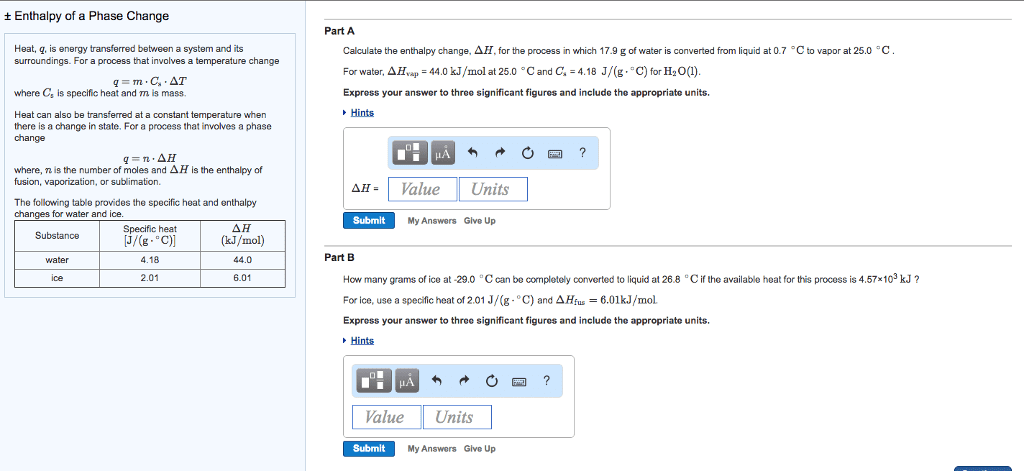
Heat, q, is energy transferred between a system and its surroundings. For a process that involves a temperature change
q=mâ Csâ ÎT
where Cs is specific heat and m is mass.
Heat can also be transferred at a constant temperature when there is a change in state. For a process that involves a phase change
q=nâ ÎH
where, n is the number of moles and ÎH is the enthalpy of fusion, vaporization, or sublimation.
The following table provides the specific heat and enthalpy changes for water and ice.
| Substance | Specific heat [J/(gâ âC)] | ÎH (kJ/mol) |
| water | 4.18 | 44.0 |
| ice | 2.01 | 6.01 |
Part A
Calculate the enthalpy change, ÎH, for the process in which 45.7 g of water is converted from liquid at 10.1 âC to vapor at 25.0 âC .
For water, ÎHvap = 44.0 kJ/mol at 25.0 âC and Cs = 4.18 J/(gâ âC) for H2O(l).
Part B
How many grams of ice at -12.5 âC can be completely converted to liquid at 8.1 âC if the available heat for this process is 5.27Ã103 kJ ?
For ice, use a specific heat of 2.01 J/(gâ âC) and ÎHfus=6.01kJ/mol.
Express your answer to three significant figures and include the appropriate units.