CHE 131 Lecture Notes - Lecture 5: Mass Number, Identity Element, Natural Abundance
CHE 131 verified notes
5/46View all
Document Summary
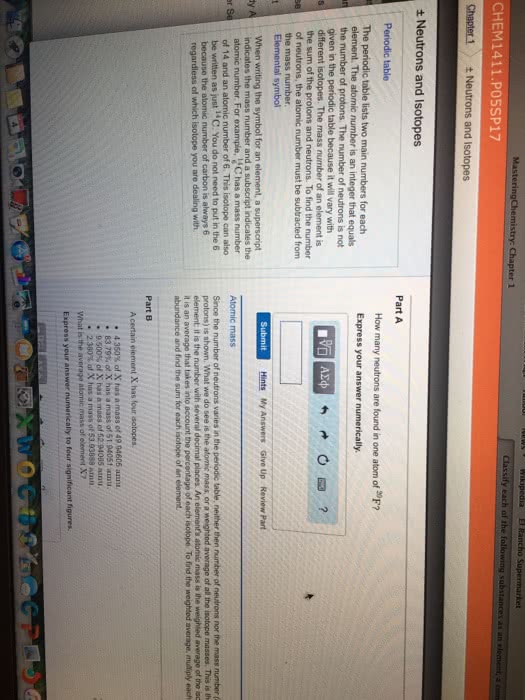
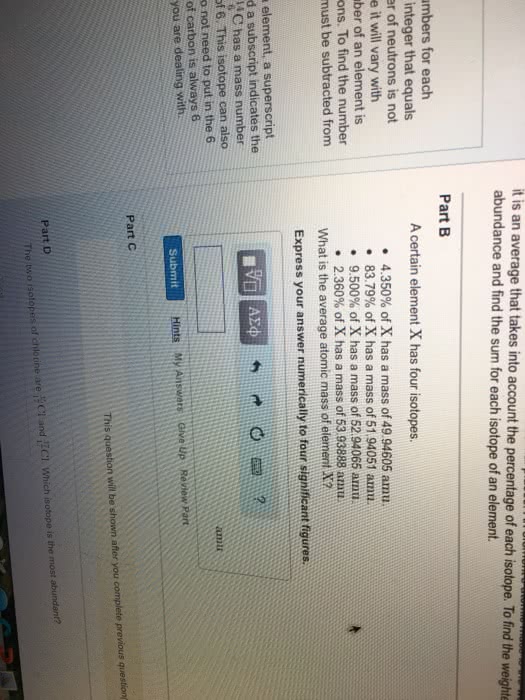
Z - atomic number (the number of protons) A - atomic mass number (the sum of protons and neutrons) In a neutral element, the number of protons = number of neutrons. Ions are formed by gaining or losing an electron. Cation - when atom loses an electron and becomes positive overall. Anion - when atom gains an electron and becomes negative overall. Isotopes are atoms of the same element with different atomic mass. Difference of the atomic mass is caused by the different number of neutrons. The neutron number has negligible effects of chemical property, but has significant nuclear properties. Isotopes of an element occur naturally at different abundances. Natural abundance - how often each isotope occurs naturally. Average atomic mass - the weighted average mass of an element and all its isotopes. The elemental mass (aka average atomic mass ) of an element can be found using the following equation.