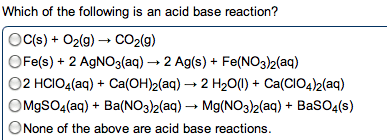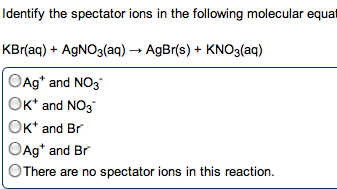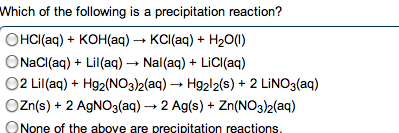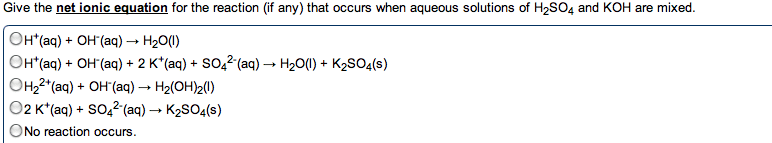CHE K121 Lecture Notes - Lecture 10: Amine, Sodium Hydroxide, Sodium Chloride
24 views2 pages
Document Summary
Reactions (acid\base: acid-base reaction (neutralization, de nitions, arrhenius: an acid is a substance that increases the concentration of h+ in water. A base is a substance that increases the oh- concentration: bronsted-lowry: an acid is a proton donor; a base is a proton acceptor (h+, strong acids: completely (or nearly completely) ionize in water. Hcl + h2o h + (aq) + cl (aq) (100%: weak acids: only partially ionize in water, strong bases: soluble hydroxides, group 1a and heavier 2a (completely dissociated in water) Ba2+ (aq) + 2oh (aq: weak bases: one type; insoluble hydroxides such as mg(oh)2. Most weak bases are ammonia derivatives (and ammonia itself: nh3 (base) react with water (acting as acid) to produce. 4 + oh : strong acid and strong base reactions. H + (aq) + cl (aq) + na+ (aq) + oh (aq) na+ (aq) + cl (aq) + h2o (l : net ionic equation: "