CHEM 001C Lecture Notes - Lecture 6: Hypobromous Acid, Acid Dissociation Constant, Ammonia
41 views2 pages
Document Summary
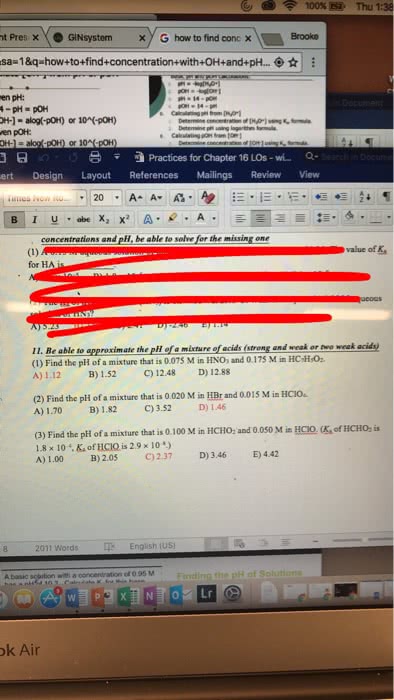
Chem 001c lecture 6: chapter 14 acid-base chemistry. Acids with pka significantly less than 1. Example: hso 3 f * sbf 5 pka = -31. K a / k b = equilibrium dissociation constants for acids and bases. B + h 2 o hb + + oh - Temperature is the only factor that can influence k value, Where k w = 1. 0 x 10 -14 at 25 according to equation: [h 3 o + ] = 10 -ph ph = -log[h 3 o + ] [oh - ] = 10 -poh poh = -log[oh - ] Example : calculate the k a of a 0. 2m aqueous solution of ch 3 ch 2 co 2 h with a ph of 4. 88. [h 3 o + ] = 10 -4. 88 = 1. 32 x 10 -5 m = x. K a = (0. 2 1. 32 x 10 ) 5 (1. 32 x 10 ) 5 2.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232