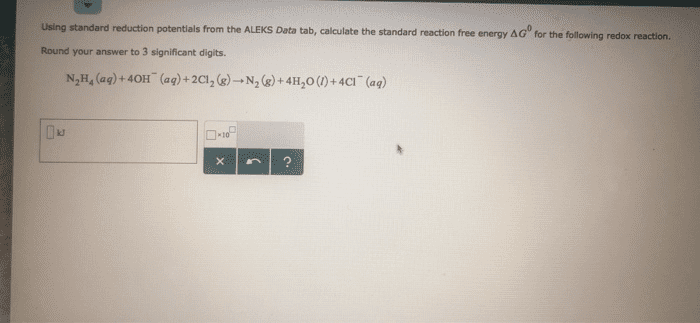
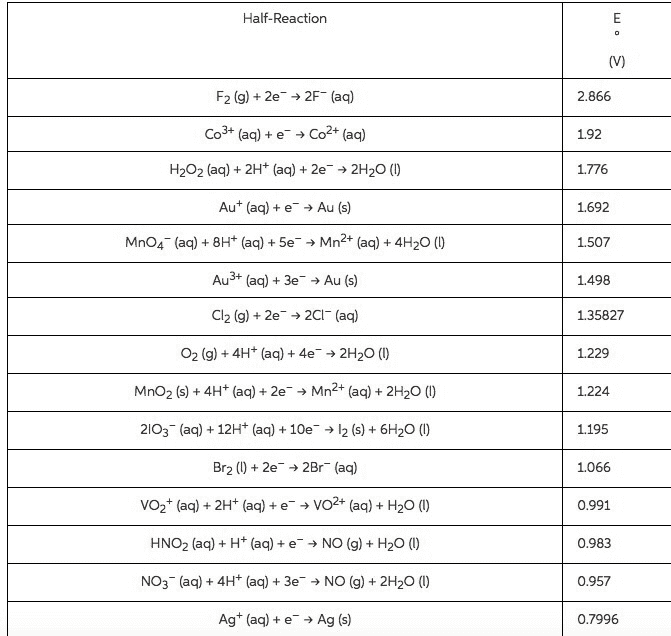
Using standard reduction potentials from the ALEKS Data tab calculate the standard reaction free energy AGO for the following redox reaction. Round your answer to 3 significant digits. N2 H (ag)+40H (aq)+2CI2 () N2 ()+4H20 (0)+4CI (aq) kJ
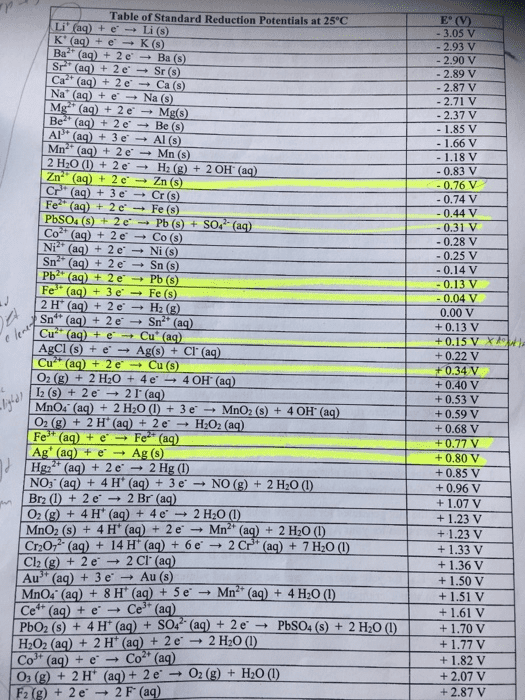
Half-Reaction 2.866 192 1.776 1692 1.507 1.498 1.35827 F2 (g)+2e >2F (aq) Co3+ (aq) + e-â Co2+ (aq) H2O2 (aq) + 2H+ (aq) + 2e-â 2H2O (1) Au+ (aq) + e-â Au (s) MnO4" (aq) + 8H+ (aq) + 5e-â Mn2+ (aq) + 4H2O (1) Au3+ (aq) + 3e-â Au (s) Cl2 (g) + 2e-â 2Cl-(aq) O2 (g)+4H+ (a)4e2H2O() MnO2 (s) + 4H+ (aq) + 2e-â Mn2+ (aq) + 2H2O (l) 21O3 (aq) +12H (aq)+10e2 (s) 6H20 () Br2)2e2Br (aq) VO2+ (aq) + 2H+ (aq) + e-â VO2+ (aq) + H2O (l) HNO2 (aq) + H+ (aq) + e-â NO (g) + H2O (l) NO3 (a) 4H+ (aq) 3e NO (g) 2H20 () Ag+ (aq) + e-â Ag (s) 1.229 1.224 1.195 1.066 0.991 0.983 0.957 0.7996
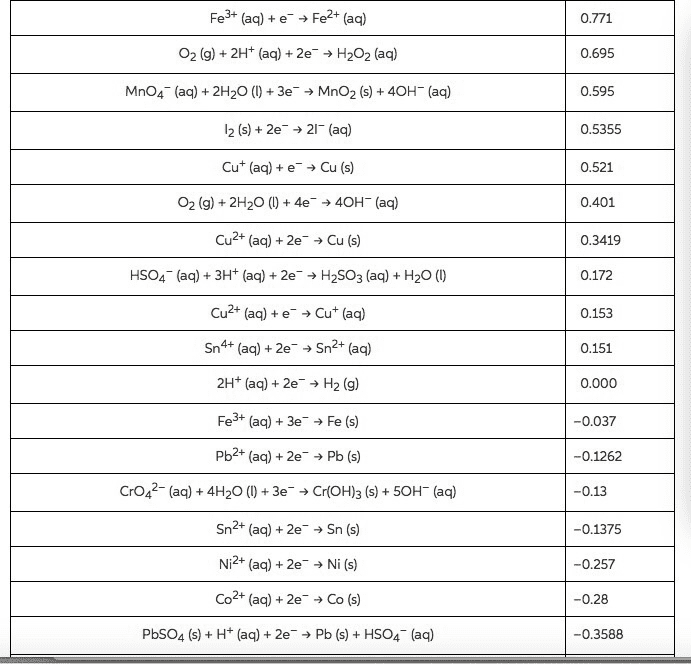
Fe3+ (aq)eFe2+ (acq) 02 (g) + 2H+ (aq) + 2e-â H2O2 (aq) MnO4 (aq) 2H2O ()3eMnO2 (s40H (aq) l2 (s)+ 2e 2 (aq) Cu (aq)Cu (s) 02 (g) + 2H2O (l) + 4e-â 4OH-(aq) Cu2+ (aq) +2e> Cu (s) HSO4 (aq)3Ht (aq)2eH2SO3 (aq)+ H2O (0) Cu2+ (aq) + e-â Cu+ (aq) Sn4+ (aq) + 2e-â Sn2+ (aq) 2H+ (aq) + 2e-â H2 (g) Fe3+ (aq) 3e Fe (s) Pb2+ (aq) + 2e-â Pb (s) CrO42-(aq)4H20( 3e Cr(OH)3 (s)+50H- (aq) Sn2+ (aq)+2e> Sn (s) Ni2+ (aq)+2eNi (s) Co2+ (aq) + 2e-â Co (s) PbSO4 (s) + H+ (aq) + 2e-â Pb (s) + HSO4-(aq) 0.695 0.595 0.5355 0.521 0.401 0.3419 0.172 0.153 0.151 0.000 -0.037 -0.1262 -0.13 -0.1375 -0.257 -0.28 -0.3588
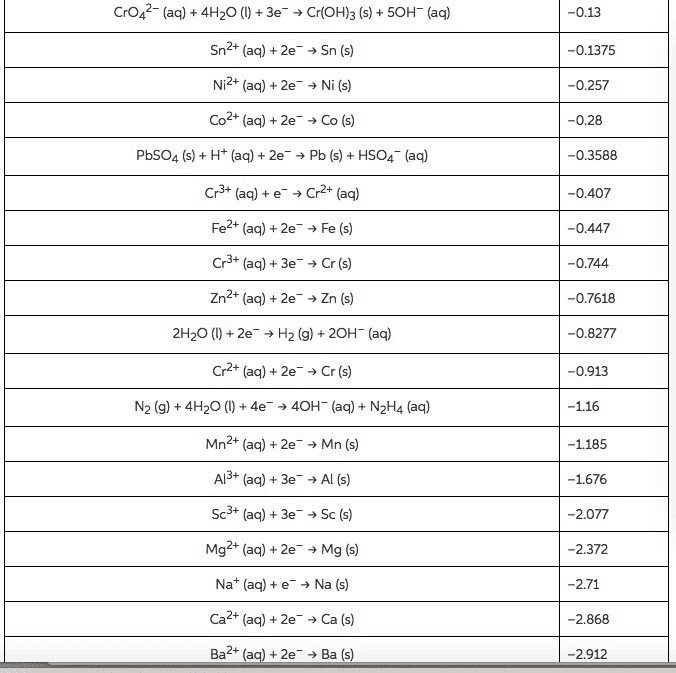
CrO42-(aq)4H20(3e Cr(OH)3 (s)+50H- (aq) Sn2+ (aq)+2e> Sn (s) Ni2+ (aq) + 2e, Ni (s) Co2+ (aq) + 2e-â Co (s) PbSO4 (s)H (aq)2ePb (sHSO4 (aq) Cr3+ (aq) + e-â Cr2+ (aq) Fe2+ (aq) +2e Fe (s) Cr3+ (aq) + 3e-â Cr(s) Zn2+ (aq)+2e Zn (s) 2H20 () 2eH2 (g) + 20H (aq) Cr2+ (aq)+2e Cr (s) N2 (g) 4H2O)+4e 40H (aq) N2H4 (aq) Mn2+ (aq)+ 2e Mn (s) Al3+ (aq)+3e Al (s) Sc3+ (aq)3eSc (s) Mg2+ (aa)+ 2e Mg (s) Nat (aq)eNa (s) Ca2+ (aq) + 2e-â Ca (s) Ba2+ (aq) + 2e-â Ba (s) -0.13 0.1375 -0.257 -0.28 0.3588 -0.407 -0.447 -0.744 0.7618 0.8277 -0.913 1.185 1.676 -2.077 2.372 2.71 2.868 2.912
Show transcribed image text Using standard reduction potentials from the ALEKS Data tab calculate the standard reaction free energy AGO for the following redox reaction. Round your answer to 3 significant digits. N2 H (ag)+40H (aq)+2CI2 () N2 ()+4H20 (0)+4CI (aq) kJ
Half-Reaction 2.866 192 1.776 1692 1.507 1.498 1.35827 F2 (g)+2e >2F (aq) Co3+ (aq) + e-â Co2+ (aq) H2O2 (aq) + 2H+ (aq) + 2e-â 2H2O (1) Au+ (aq) + e-â Au (s) MnO4" (aq) + 8H+ (aq) + 5e-â Mn2+ (aq) + 4H2O (1) Au3+ (aq) + 3e-â Au (s) Cl2 (g) + 2e-â 2Cl-(aq) O2 (g)+4H+ (a)4e2H2O() MnO2 (s) + 4H+ (aq) + 2e-â Mn2+ (aq) + 2H2O (l) 21O3 (aq) +12H (aq)+10e2 (s) 6H20 () Br2)2e2Br (aq) VO2+ (aq) + 2H+ (aq) + e-â VO2+ (aq) + H2O (l) HNO2 (aq) + H+ (aq) + e-â NO (g) + H2O (l) NO3 (a) 4H+ (aq) 3e NO (g) 2H20 () Ag+ (aq) + e-â Ag (s) 1.229 1.224 1.195 1.066 0.991 0.983 0.957 0.7996
Fe3+ (aq)eFe2+ (acq) 02 (g) + 2H+ (aq) + 2e-â H2O2 (aq) MnO4 (aq) 2H2O ()3eMnO2 (s40H (aq) l2 (s)+ 2e 2 (aq) Cu (aq)Cu (s) 02 (g) + 2H2O (l) + 4e-â 4OH-(aq) Cu2+ (aq) +2e> Cu (s) HSO4 (aq)3Ht (aq)2eH2SO3 (aq)+ H2O (0) Cu2+ (aq) + e-â Cu+ (aq) Sn4+ (aq) + 2e-â Sn2+ (aq) 2H+ (aq) + 2e-â H2 (g) Fe3+ (aq) 3e Fe (s) Pb2+ (aq) + 2e-â Pb (s) CrO42-(aq)4H20( 3e Cr(OH)3 (s)+50H- (aq) Sn2+ (aq)+2e> Sn (s) Ni2+ (aq)+2eNi (s) Co2+ (aq) + 2e-â Co (s) PbSO4 (s) + H+ (aq) + 2e-â Pb (s) + HSO4-(aq) 0.695 0.595 0.5355 0.521 0.401 0.3419 0.172 0.153 0.151 0.000 -0.037 -0.1262 -0.13 -0.1375 -0.257 -0.28 -0.3588
CrO42-(aq)4H20(3e Cr(OH)3 (s)+50H- (aq) Sn2+ (aq)+2e> Sn (s) Ni2+ (aq) + 2e, Ni (s) Co2+ (aq) + 2e-â Co (s) PbSO4 (s)H (aq)2ePb (sHSO4 (aq) Cr3+ (aq) + e-â Cr2+ (aq) Fe2+ (aq) +2e Fe (s) Cr3+ (aq) + 3e-â Cr(s) Zn2+ (aq)+2e Zn (s) 2H20 () 2eH2 (g) + 20H (aq) Cr2+ (aq)+2e Cr (s) N2 (g) 4H2O)+4e 40H (aq) N2H4 (aq) Mn2+ (aq)+ 2e Mn (s) Al3+ (aq)+3e Al (s) Sc3+ (aq)3eSc (s) Mg2+ (aa)+ 2e Mg (s) Nat (aq)eNa (s) Ca2+ (aq) + 2e-â Ca (s) Ba2+ (aq) + 2e-â Ba (s) -0.13 0.1375 -0.257 -0.28 0.3588 -0.407 -0.447 -0.744 0.7618 0.8277 -0.913 1.185 1.676 -2.077 2.372 2.71 2.868 2.912