CHM 132 Study Guide - Midterm Guide: Ludwig Boltzmann, Sustainable Energy, Coffee Cup
Document Summary
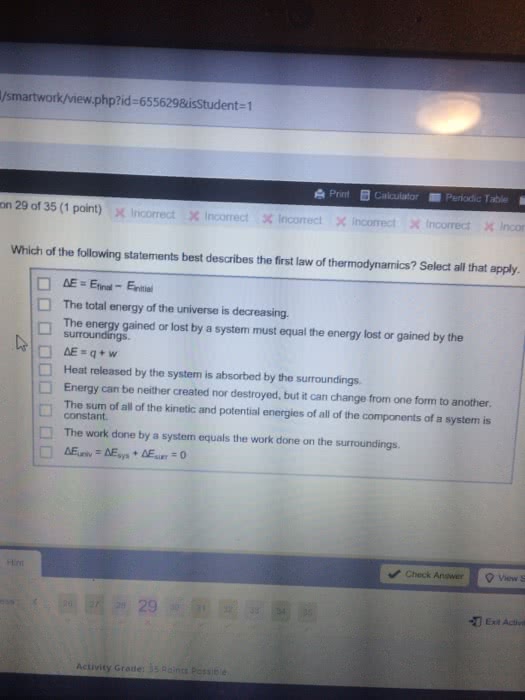
Energy the capacity to produce and effect. Potential energy energy due to position or composition. Kinetic energy energy associate with movement. E = ef ei = q + w: e = qv. Heat t(cid:396)a(cid:374)sfe(cid:396)/ flo(cid:449) of e(cid:374)e(cid:396)gy as a (cid:396)esult of t. Work a force acting over a distance (w = -p v(cid:895) State function path independent (e, h, s, g, p, v) Non-state function not path independent (q, w) 1st law of thermodynamics: energy of the universe is constant. Cannot be destroyed or created: euniv = (cid:1004) = esys + esurr. Exothermic heat is lost from the system and enters the surroundings (q < 0) Endothermic heat is gained by the system and is lost from the surroundings (q > 0) Note: when finding work, make sure to multiply the atm*l result by 101. 3j/atm*l. if you do not, then it is not in j and cannot be used. Hwith limiting reagents = qp/ moles made.