CHE 101 Chapter Notes - Chapter 4: Collision Frequency, Acid Hydrolysis, Arrhenius Equation
Document Summary
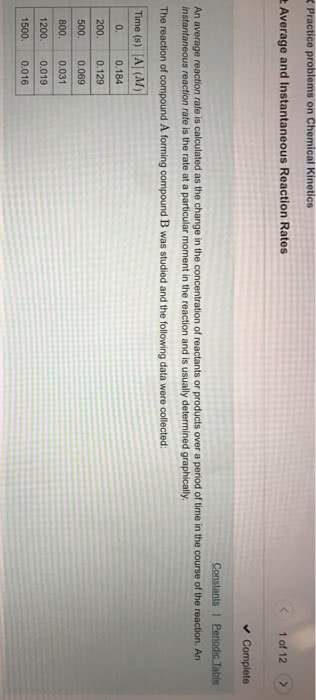
Rate of change of extent of reaction is the rate of reaction. Rate of reaction is positive for product and negative for reactant. Rate =1/b( [b]/ t) = -1/a ( [a]/ t) It goes on decreasing as the reaction progress due to decrease in the concentration(s) of the reactant(s). Unit of rate of reaction : mol l -1 s -1. The rate measured over a long time interval is called average rate and the rate measured for an infinitesimally small time interval is called instantaneous rate. In a chemical change, reactants and products are involved. As the chemical reaction proceeds, the concentration of the reactants decreases, i. e. , products are produced. The rate of reaction (average rate) is defined as the change of concentration of any one of its reactants (or products) per unit time. For reaction aa + bb + cc+ . It is the reaction which completes in a single step.