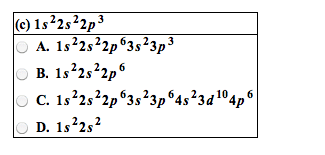CHEM10007 Lecture Notes - Lecture 5: Ionic Compound, Chemical Bond, Covalent Bond

LECTURE 5 - CHEMICAL BONDING, IONS & IONIC
COMPOUNDS
CHEMICAL BONDING
•Chemical bond: the attraction that holds atoms together in chemical compounds.
•Different compounds may have different types of chemical bonds.
TWO TYPES OF CHEMICAL BOND
•Covalent bonds: atoms joined together by
shared electrons.
•Ionic bonds: attraction between oppositely
charged ions.
•An ion is an atom or group of atoms with a
net positive or negative charge.
NaCl and HCl
•NaCl is a white crystalline solid at room
temperature whilst HCl is a pungent,
corrosive gas.
•Properties are linked to the type of chemical
bond.
•NaCl is a typical ionic substance.
•HCl is a molecular substance with a covalent
bond between the atoms.
•Na is one of the most reactive metals.
•Cl is a diatomic gas.
FORMATION OF IONS
•An electron is transferred between the atoms.
•Both ions end up with a noble gas configuration.
IONIC BONDING
•In sodium chloride, sodium (Na+) and chloride (Cl- ) ions form in equal numbers.
•Positive and negative ions have opposite charge and attract each other.
•An ionic solid is formed.
SOLID SODIUM CHLORIDE
•NaCl forms a cubic structure.
•Each Na+ cation is surrounded by 6 Cl- anions and each Cl- anion by 6
Na+ cations.
Document Summary
Lecture 5 - chemical bonding, ions & ionic. Chemical bonding: chemical bond: the attraction that holds atoms together in chemical compounds, different compounds may have different types of chemical bonds. Two types of chemical bond: covalent bonds: atoms joined together by shared electrons, ionic bonds: attraction between oppositely charged ions, an ion is an atom or group of atoms with a net positive or negative charge. Formation of ions: an electron is transferred between the atoms, both ions end up with a noble gas configuration. Ionic bonding: in sodium chloride, sodium (na+) and chloride (cl- ) ions form in equal numbers, positive and negative ions have opposite charge and attract each other, an ionic solid is formed. Solid sodium chloride: nacl forms a cubic structure, each na+ cation is surrounded by 6 cl- anions and each cl- anion by 6. Chemical formulae: ionic compounds result when metals react with nonmetals.