CHEM1011 Lecture Notes - Lecture 18: Intermolecular Force, Surface Tension, Joule

Lecture 18: Intermolecular forces
Intramolecular forces
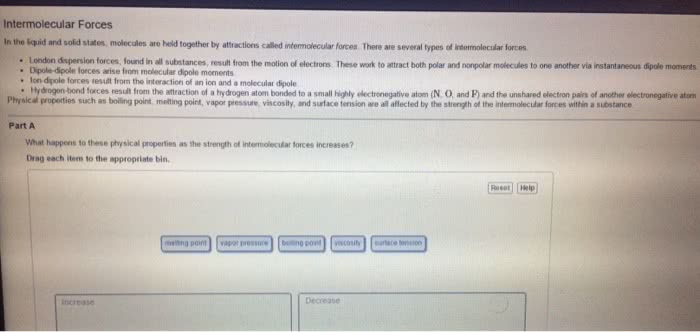
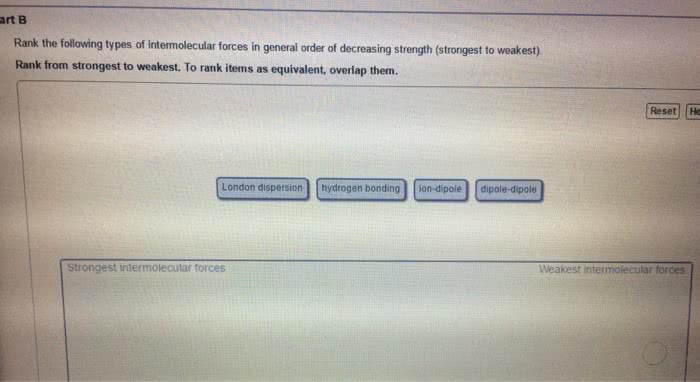
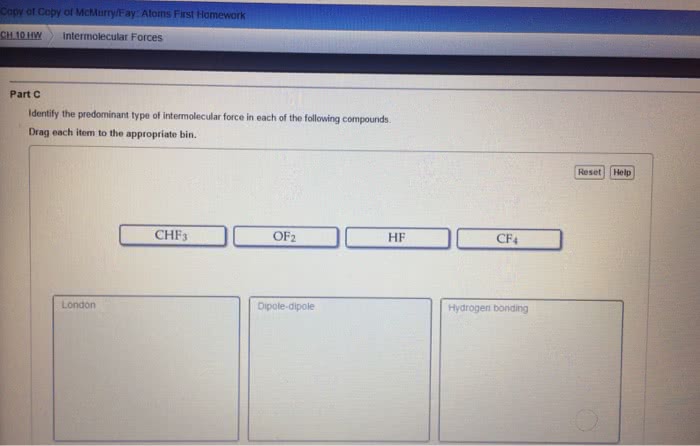
Forces between molecules
- Responsible for differences in some physical properties such as boiling
point and surface tension
- Three main types
Dipole – dipole forces
Dispersion forces
Hydrogen bonds
Dipole- dipole forces
A molecule has a dipole moment if
- It contains polar bonds
- Is asymmetric in shape
- There is a attratio etwee the δ+ of oe oleule ad δ – of another molecule.
- Typical energy 5 – 25 kJ mol–1
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
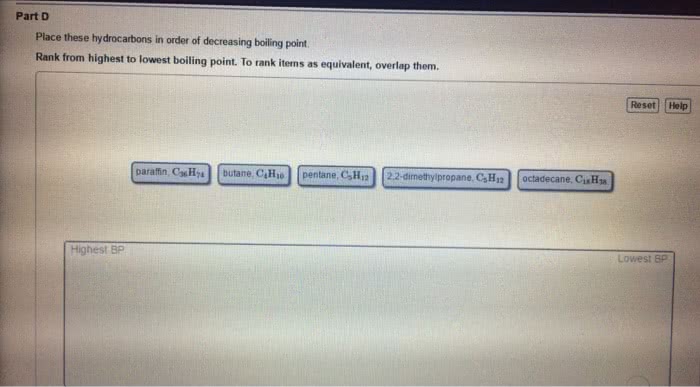
Responsible for differences in some physical properties such as boiling point and surface tension. There is a(cid:374) attra(cid:272)tio(cid:374) (cid:271)etwee(cid:374) the + of o(cid:374)e (cid:373)ole(cid:272)ule a(cid:374)d of another molecule. Typical energy 5 25 kj mol 1. Electric fields influence charge of other molecules (see pic below) Typical energy of dispersion forces is 0. 1 40 kj mol 1 (much less than a covalent bond). One direct measure of the strength of the intermolecular forces is the boiling point of a substance. When the molecular weights are similar, then dipole-dipole interactions can strongly influence the boiling point. Higher boiling point= bigger sa and stronger interactions. More exposed you make oxygen, more unbalanced you make oxygen, higher dipole moment, higher boiling point. The force increases in strength with molecular mass. Forces associated with permanent dipoles are found only in substances with overall dipole moments (polar molecules). When comparing substances of widely different masses, dispersion forces are usually more significant than dipolar forces.