CEM 141 Lecture Notes - Lecture 30: Trigonal Pyramidal Molecular Geometry, Lone Pair, Vsepr Theory
Document Summary
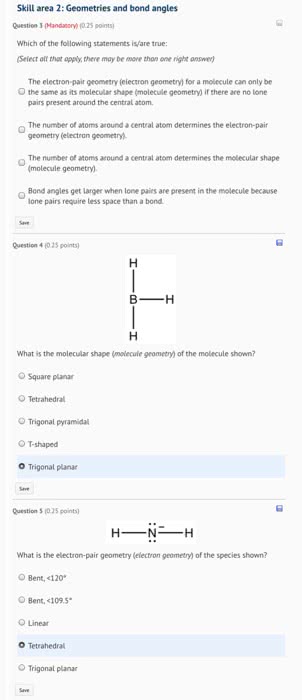
Electron center geometry is not (always) the same as molecular shape. To determine the shape of a molecule, consider only the atoms, not the lone pairs. Example: a molecule with 4 centers of electron density form a tetrahedral geometry. If all 4 centers are bonds than the geometry stays tetrahedral, but the shape is also tetrahedral. If one of those bonds is a lone pair, the shape is trigonal pyramidal. If two of those bonds are lone pairs, the shape is bent. = # of protons - # of core electrons. Increases down a row (as atomic radius gets smaller), stays same down a column (as atomic radius gets larger) Ability of an element to attract electrons to itself in a bond. Example: out of f, cl, br, and i, f is the smallest and has the strongest attractive force between the electrons and the nucleus.