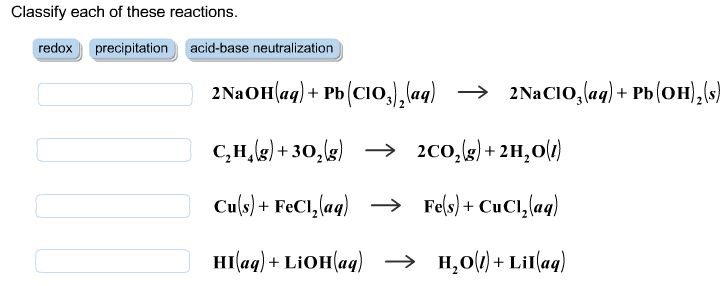CHE 2A Lecture Notes - Lecture 7: Barium Chloride, Electrolyte, Chemical Equation

19
CHE 2A Full Course Notes
Verified Note
19 documents
Document Summary
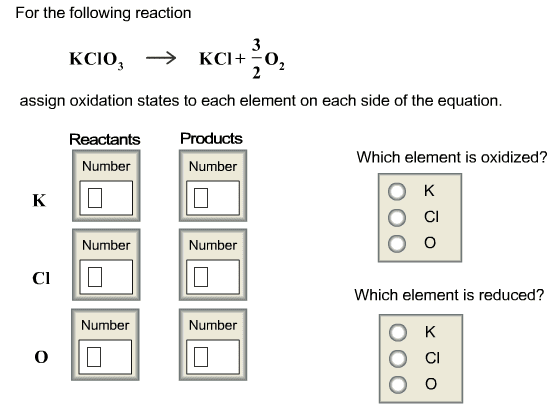
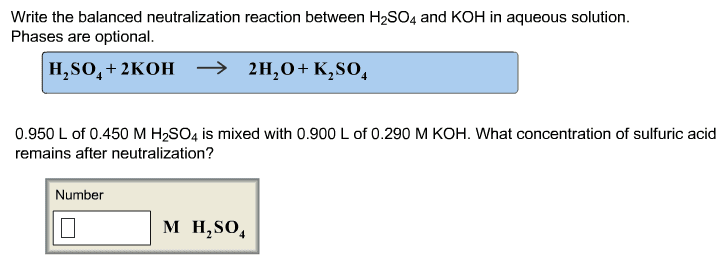
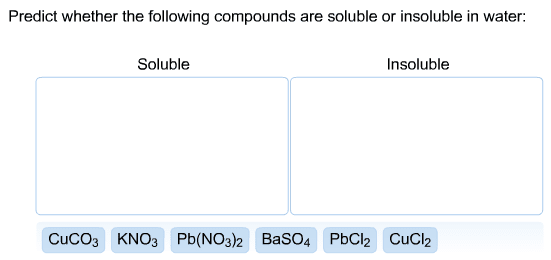
V1 x m1 = v2 x m2 ex) what volume of 15. 8m hno3 is required to make 250 ml of a 6. 0m solution? (15. 8 m)(x ml) = (250 ml)(6. 0 m) 15. 8x = 1500 x = 1500/15. 8 x=94. 9 ml hno3. A + bc ac + b . Single displacement reactions: leo = loss of electrons, oxidized, ger= gain of electrons, reduction. Reactions: cations change partners, formulas of products are determined by the chares of the reactant ions, occur only if they form a weak electrolyte or non-electrolyte i. e. bacl2(aq) + na2co3(aq) baco3(s) + 2nacl(aq) Molecular equations: balanced, shows states, all substances electrically neutral i. e. agno3(aq) + kcl(aq) agcl(s) + kno3(aq) Ionic equation: balanced, shows states, shows strong electrolytes at dissociated ions, net charge balance i. e. ag+ (aq) + no3 - (aq) + k+ (aq) + cl- (aq) agcl(s) + k+ (aq) + no3 - (aq)