CHEM 14A Lecture Notes - Lecture 7: Wolfgang Pauli, Coordinate Space, Atomic Orbital
21 views3 pages
17 Jun 2019
School
Department
Course
Professor
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
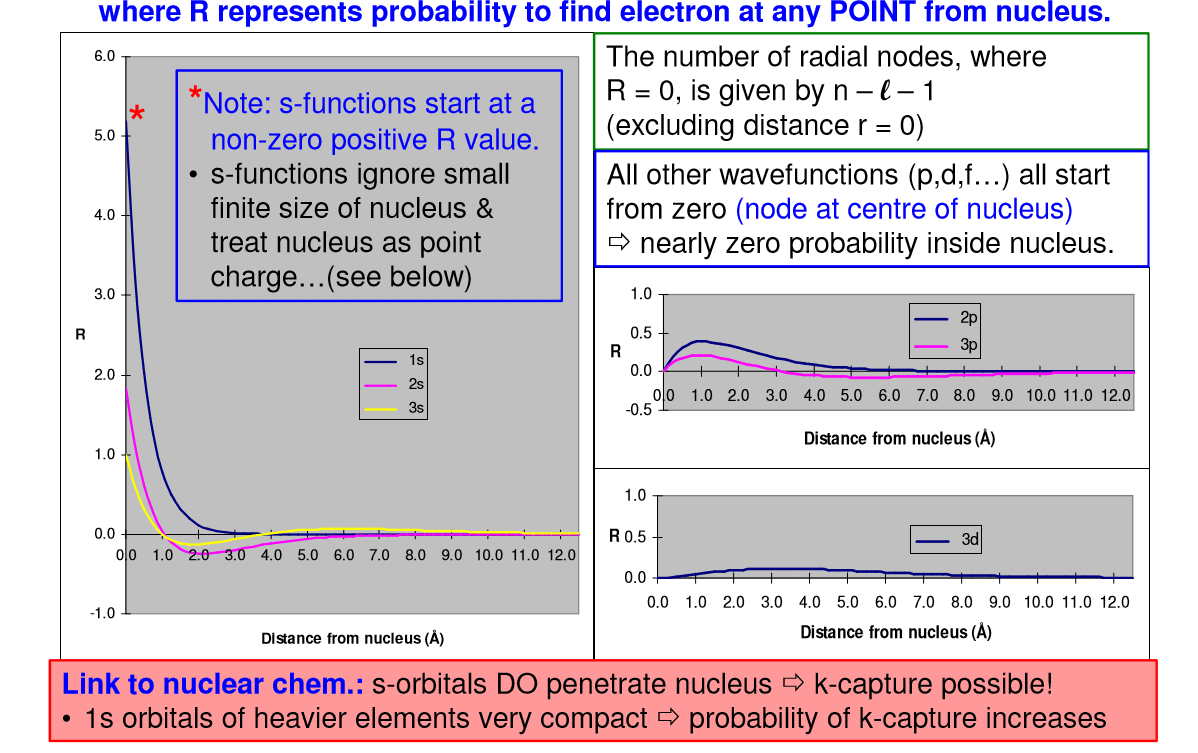
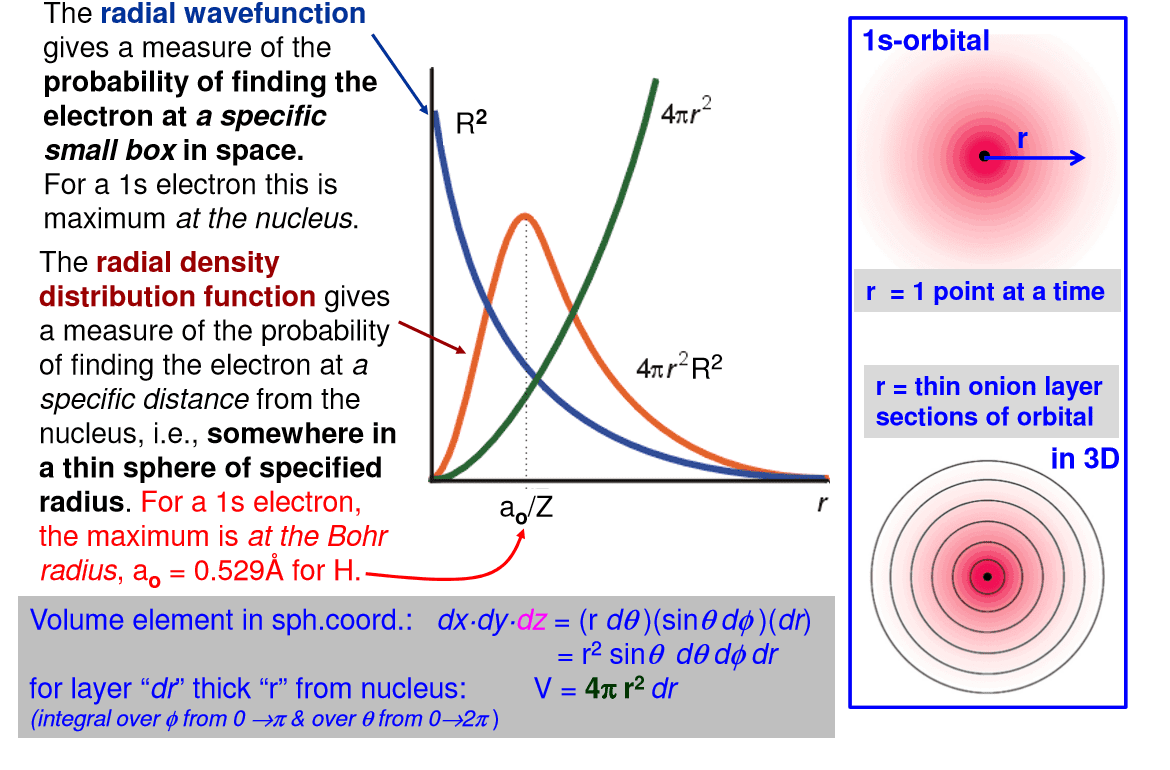
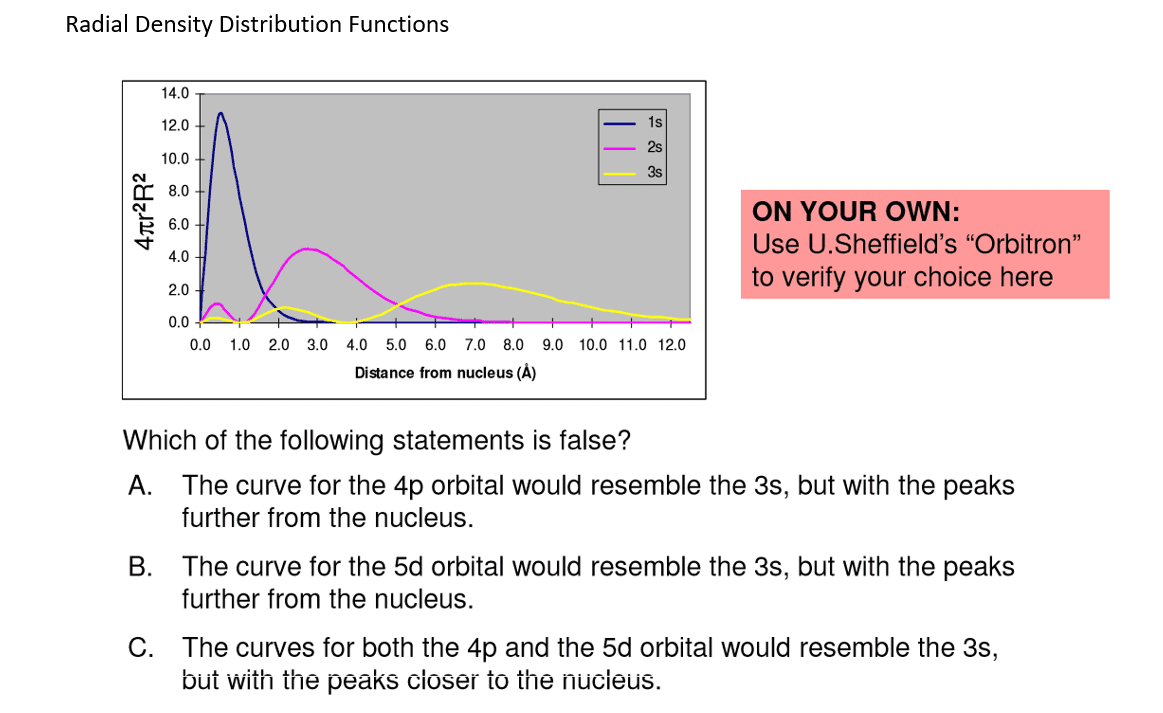
The probability of finding an electron at a point in an atom is referred to as the probability density (Ï2). The spatial distribution of these densities can be derived from the radial wave function R(r) and angular wave function Y(θ,Ï), then solving the Schrödinger equation for a specific set of quantum numbers.
Which of the following statements about nodes and probability density are accurate?
| a-The 2s orbital does not have any nodes. | |
| b-The probability of finding an electron at the center of a p orbital is zero. | |
| c-The 3p orbitals have two nodes. | |
| d-The probability of finding an electron at the center of a d orbital is greater than zero. | |
| e-The 4f orbitals have three nodes. |
alkalosis homeostasis cryogenesis |
carbohydrates lipids nucleic acids |
water is adhesive water istransparent water expands when itfreezes |
reaction initiation activation |
cytoplasm mitochondria nucleus |
oxygen and sugars proteins and sugars carbon dioxide andoxygen |
pigments, products products, reactants elements, atoms |
ATP starch glycogen |
fats steroids amino acids |
Kreb's cycle acetyl-CoAformation electron transportchain |
fermentation the Kreb cycle the electron transportchain |
Cool the reactants. Decrease the pH. Add an enzyme(catalyst). |
nucleus mitochondria a cell membrane |
cell walls chromosomes mitochondria |
osmosis active transport passive transport |
ribosome mitochondrion cytoskeleton |
diffusion active transport passive transport |
lipids proteins carbohydrates |
neither take up water orlose water take up water andswell transport water into thecell by active transport. |
lipid storage DNA replication photosynthesis |
chromosome movementduring mitosis protein synthesis ATP production |
metaphase anaphase telophase |
metaphase anaphase telophase |
metaphase anaphase telophase |
Synthesis phase (S) G1 cytokinesis |
4 daughter cells, 5chromosomes each 2 daughter cells, 10chromosomes each 2 daughter cells, 5chromosomes each |
10 20 Not enough informationprovided |
the number of chromosomesallotted to each daughter cell must be halved sister chromatids must beseparated into different daughter cells it produces new arrays ofalleles on chromosomes and contributes to genetic diversity |
are in Interphase are in metaphase are undergoingMeiosis |
twice as many chromosomesas the original cell an independent assortmentof the original cell's chromosomes an identical copy of theoriginal cell's chromosome. |
A-C-A-G-C-C-G-T-A T-G-T-C-G-G-C-A-T U-G-U-C-G-G-C-A-U |