CHEM 104 Lecture Notes - Lecture 9: Gas Constant, Arrhenius Equation, Elementary Reaction
33 views6 pages
Document Summary
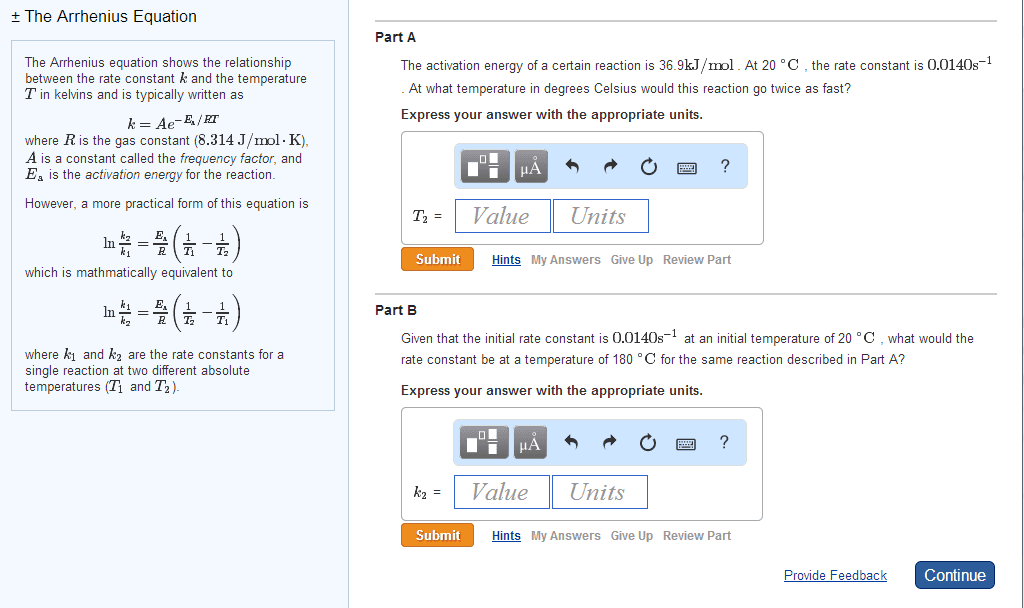
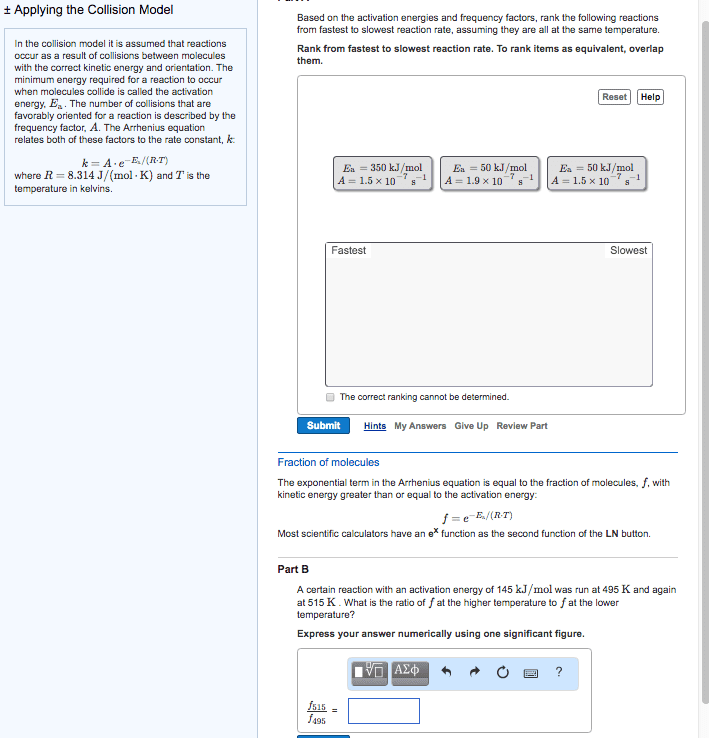
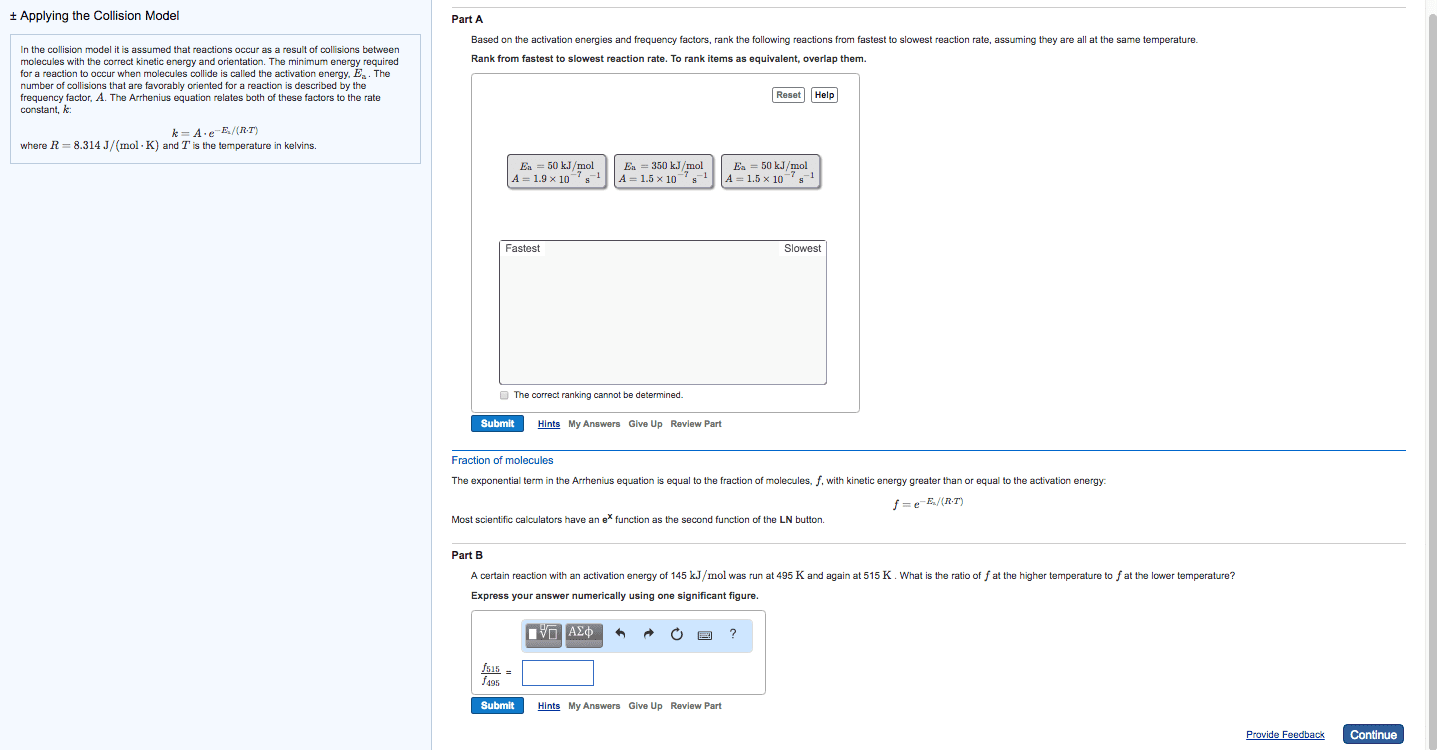
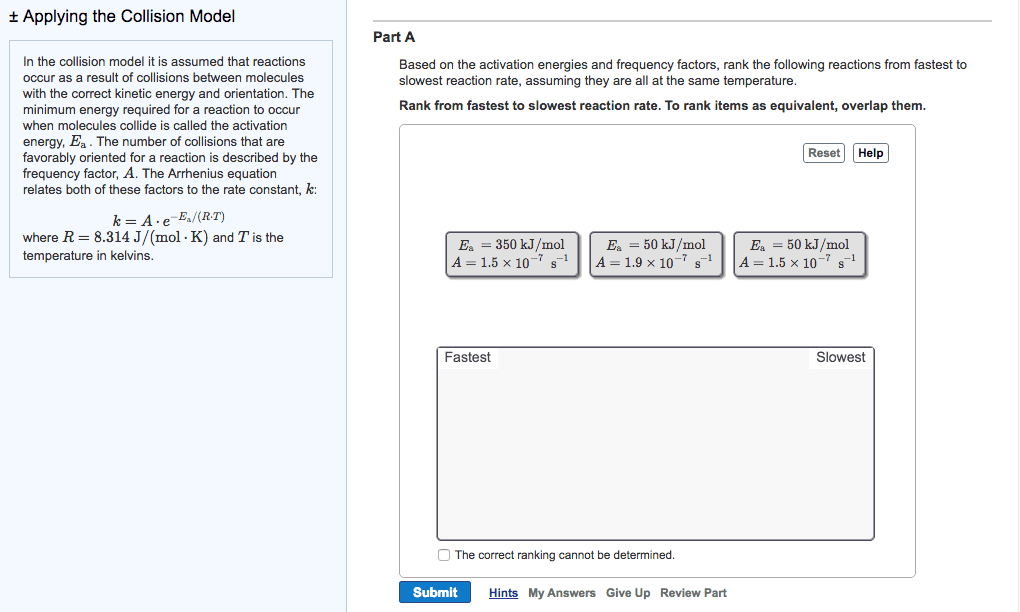
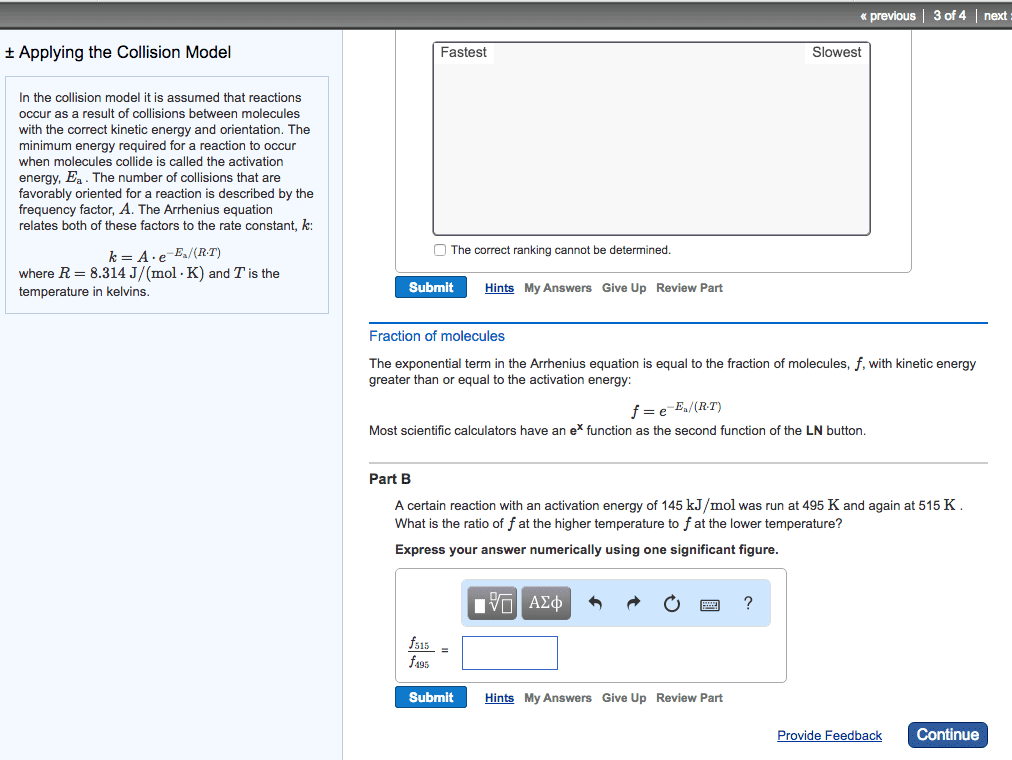
At a higher temperature a greater fraction of reactant molecules have enough energy to surmount the activation energy barrier. A rxn is faster at a higher temperature because its rate constant is larger. A rate constant is constant only for a given reaction at a given temperature k = ae-ea/rt. A = frequency factor e = base of the natural log. R = ideal gas law constant (8. 314 j/molk) * as ea increase the reaction becomes slower * Activation energy and frequency can be obtained from experimental measurements of rate constants a function of temperature. Activation energy can be modified to be linear by taking the natural logs of both sides k = ae-ea/rt. Ln (k) = ln (a) + ln (e-ea/rt) = ln (a) + (-ea/rt) Ln (k) = ear x 1t + ln (a) * temperature and activation rate are exponentially related. * reaction rete and ea are exponentially related.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232