CHEM 1110 Midterm: Chemistry 1110 2009 Fall Test 3
Document Summary
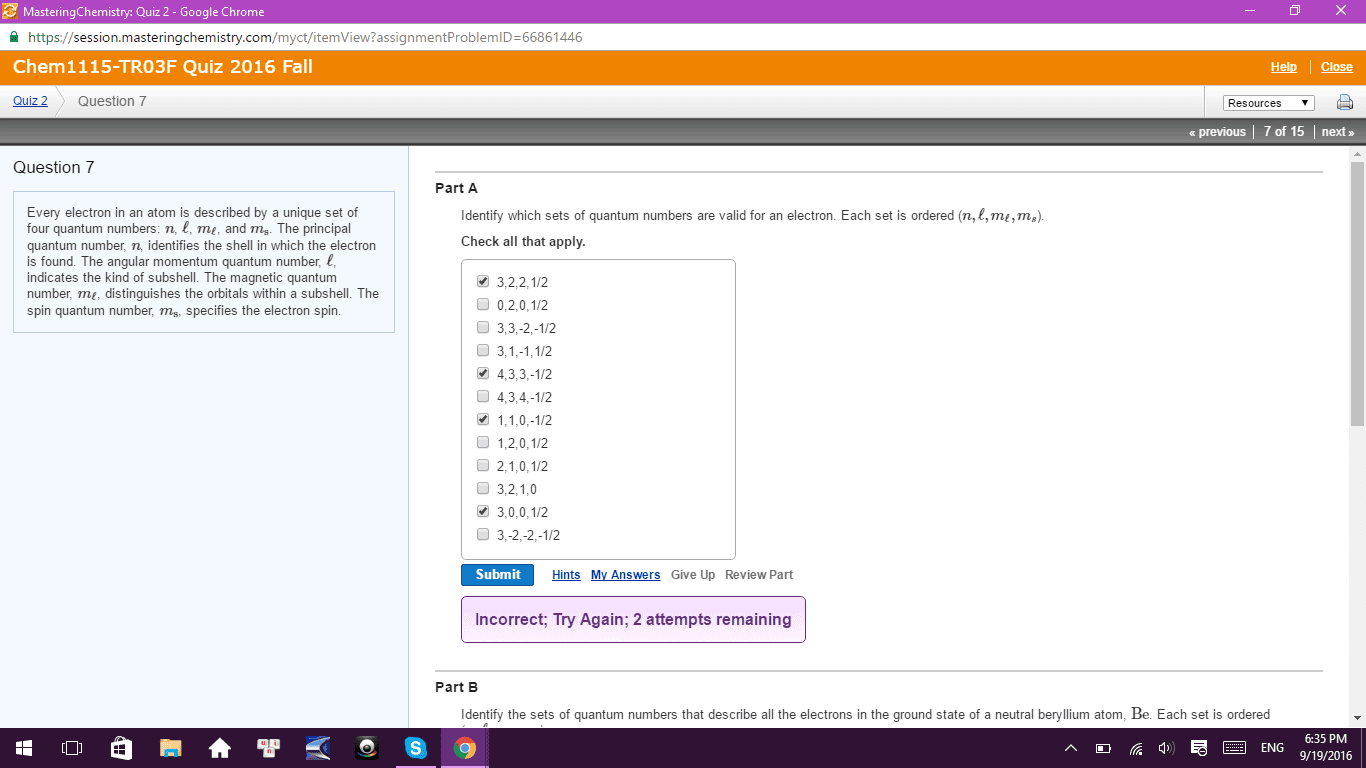
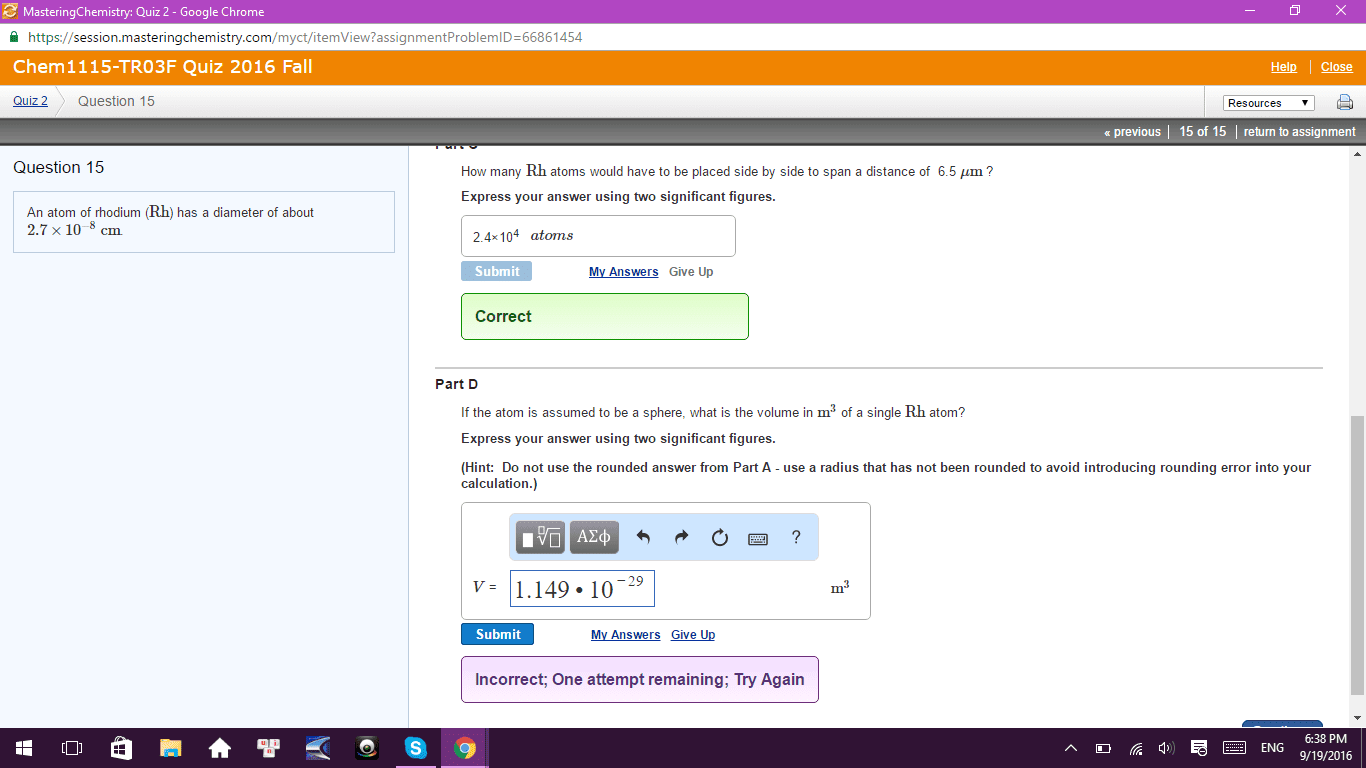
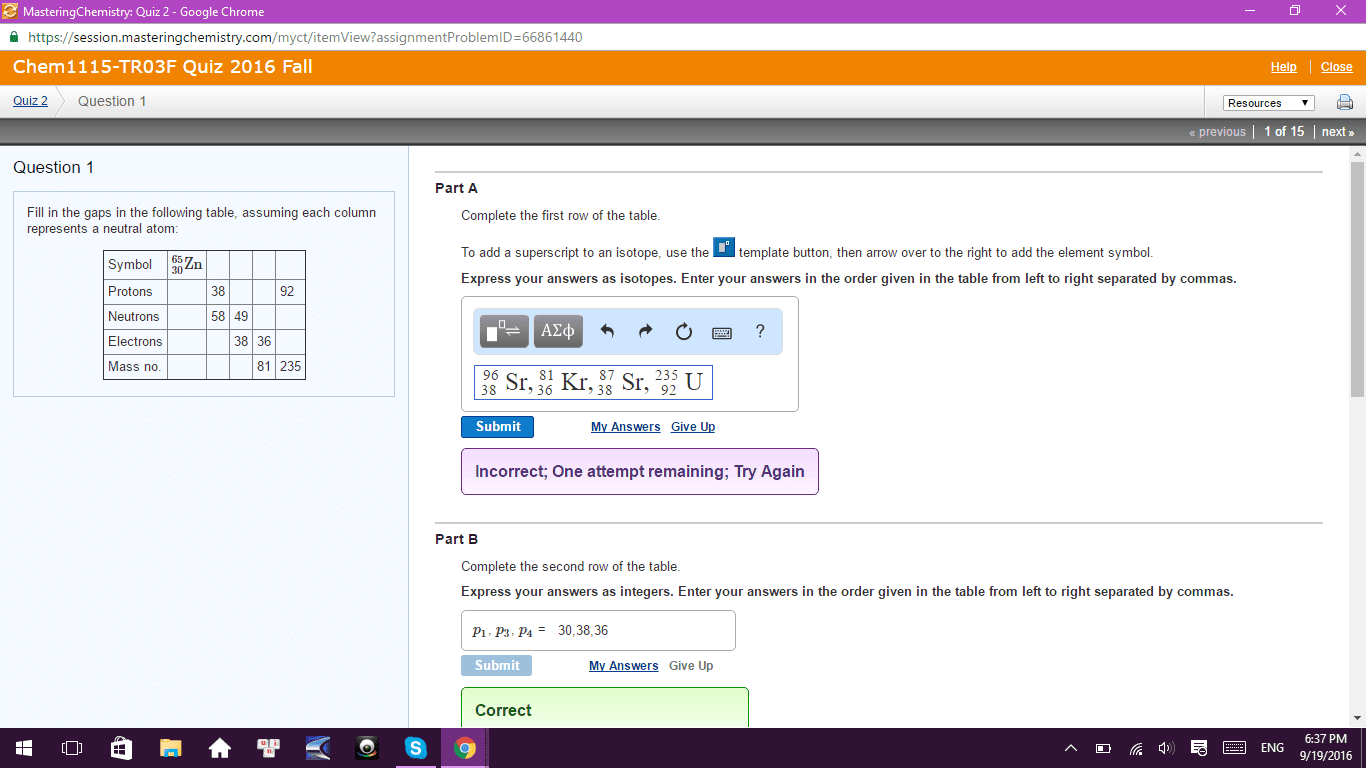
Test 3a: multiple choice (48 pts): clearly indicate the correct answer in the space provided. The notation for the subshell with n = 5 and l = 3 is: 5d subshell, 5p subshell, 5f subshell, 5g subshell, 5s subshell. "no two electrons in the same atom can have all their quantum numbers the same. " This statement is based on the work of: de broglie, heisenberg, einstein, pauli, schr dinger. A correct description for the electron configuration of an iron atom is: [ar] 3s2 3d6, paramagnetic, [ar] 4s2 3d6, diamagnetic, [ar] 4s1 3d7, paramagnetic, [ar] 3d8, paramagnetic, [ar] 4s2 3d6, paramagnetic. 3s 3p: [ne] , [ne] , [ne] , [ne] , [ne] . Which atom has the smallest radius: ca, ba, k, mg, c. Which atom has the largest first ionization energy: al, sr, ga, cr, fr. What is the correct electron configuration for cu+2: 1s22s23s23p64s13d10, 1s22s23s23p64s23d7, 1s22s23s23p63d9, 1s22s23s23p64s23d9, none of these.