CHEM 104 Study Guide - Midterm Guide: Enantiomer, Benzene, Chemical Formula
Document Summary
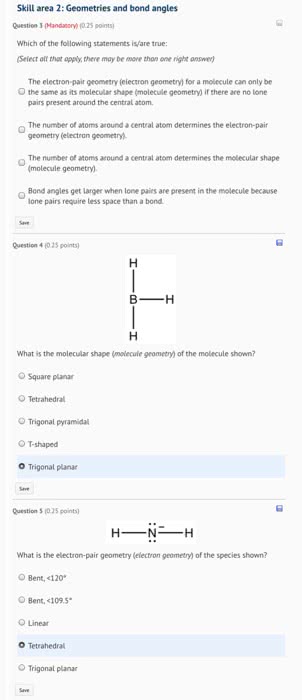
Used to determine structure around a central atom. Electron region geometry: geometry of the atoms or lone pairs of electrons connected to the central atom. Single, double, and triple bonds count as 1 electron region. Each lone pair of electrons counts as 1 electron region. Molecular geometry: geometry of the atoms around the central atom (does not include the lone pairs) Chart showing vsepr models (only molecular geometry for electron regions 2-4 are shown because those are the ones we deal with for organic chemistry) In covalent bonds atoms share electrons but the sharing is not always equal because some elements are more electronegative than others so they attract the electrons more to themselves. Electronegativity is the ability of atoms in a molecule to attract electrons to themselves. The greater the difference in electronegativity of the atoms involved in the bond the more polar the bond is.