CHEM 14A Chapter Notes - Chapter 2B.3: Electronegativity, Formal Charge, Lewis Structure
Document Summary
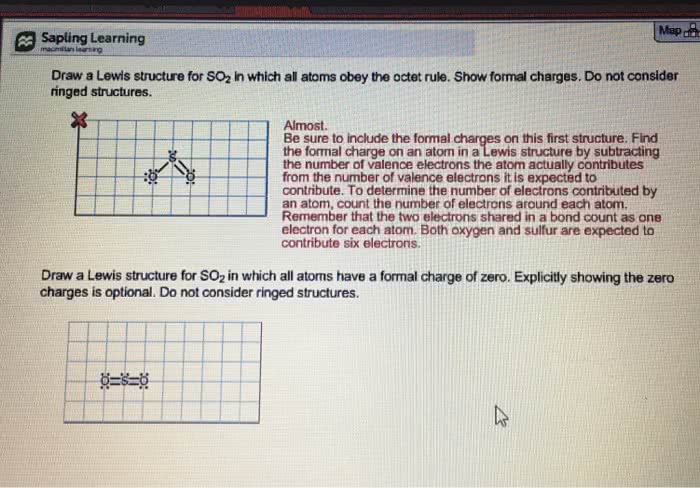
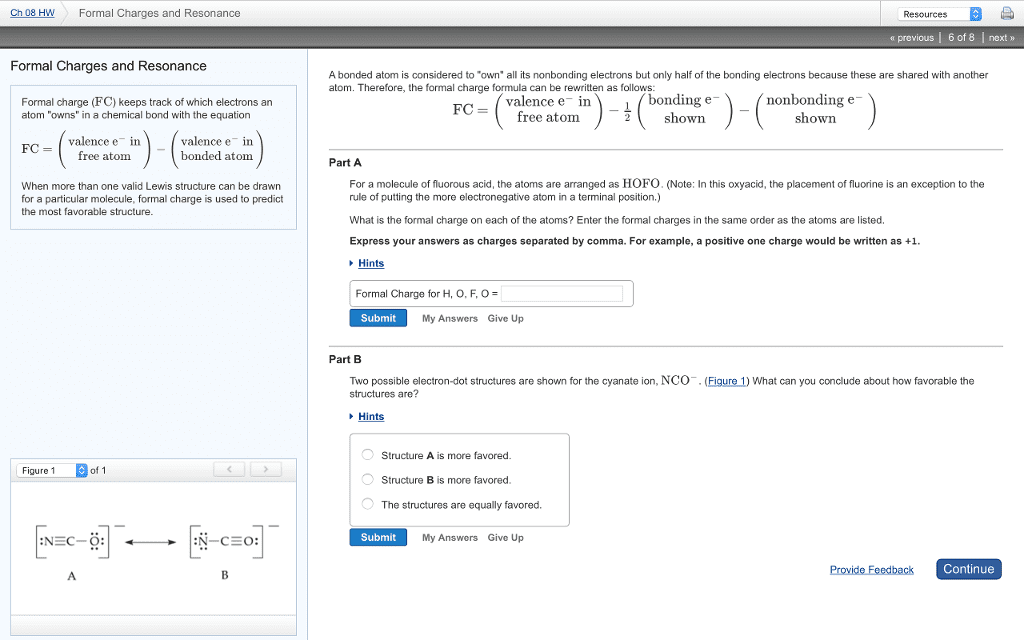
Nonequivalent lewis structures do not in general make the same contribution to a resonance structure. To decide which structures are likely to make the major contribution is to compare the number of valence electrons distributed around each atom in a structure with the number of valence electrons distributed on the free atom. Smaller difference -> lower energy -> greater contribution. Formal charge: a measure of redistribution of electrons. The charge an atom would have if the bonding were perfectly covalent in the sense that the atom had exactly half-share in each pair of bonding electrons. V: number of valence electrons in the free atom. L: number of electrons present on the bonded atom as lone pairs. B: number of bonding electrons on the atom. If the atom has more electrons in the molecule than when it"s free and neutral, the atom has a negative formal charge. The opposite would have a positive formal charge.