CBMS107 Lecture Notes - Lecture 3: Valence Electron, Noble Gas, Ionic Bonding
Document Summary
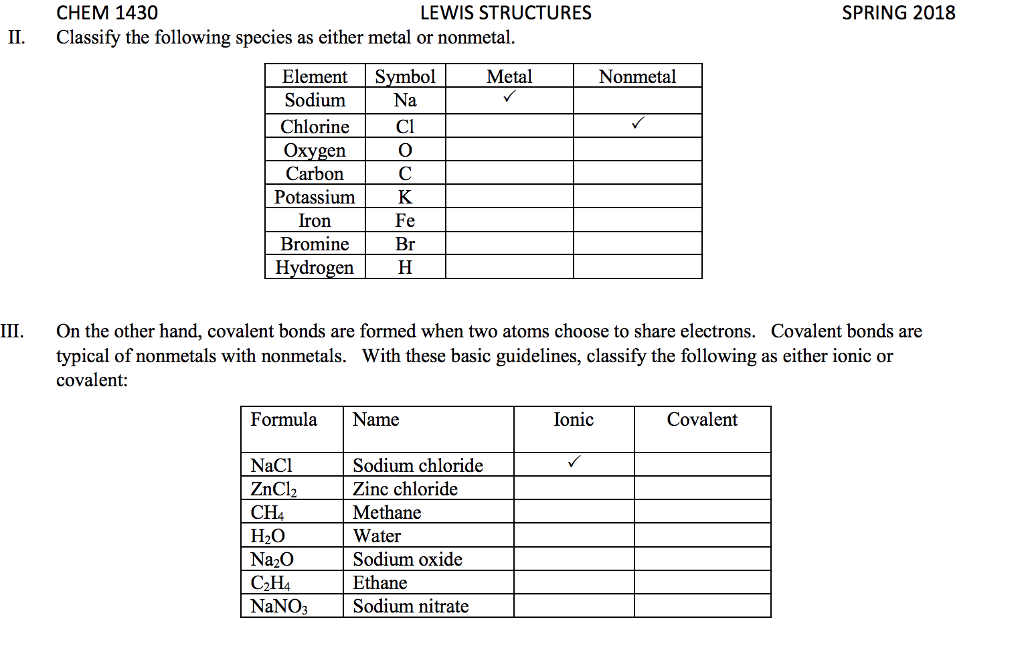
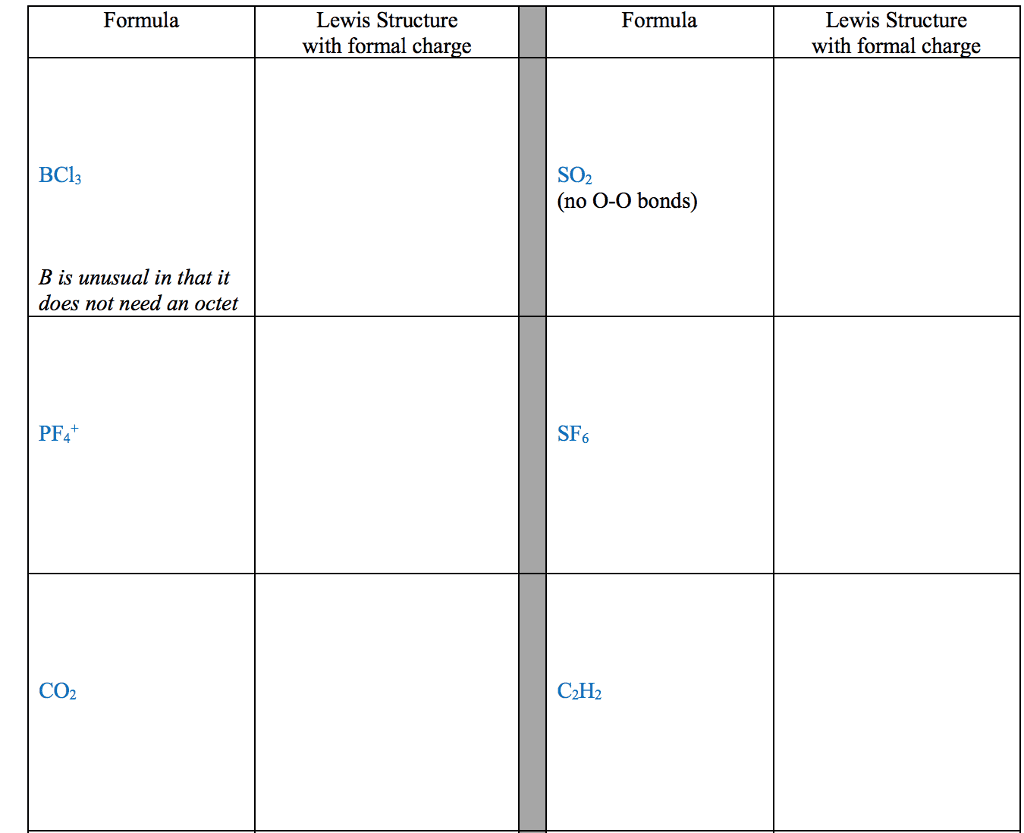
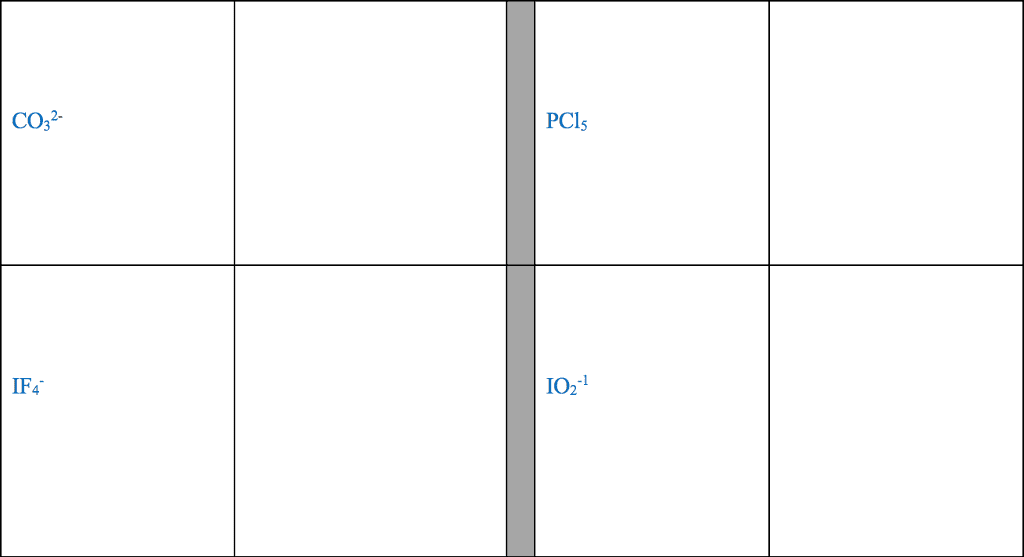
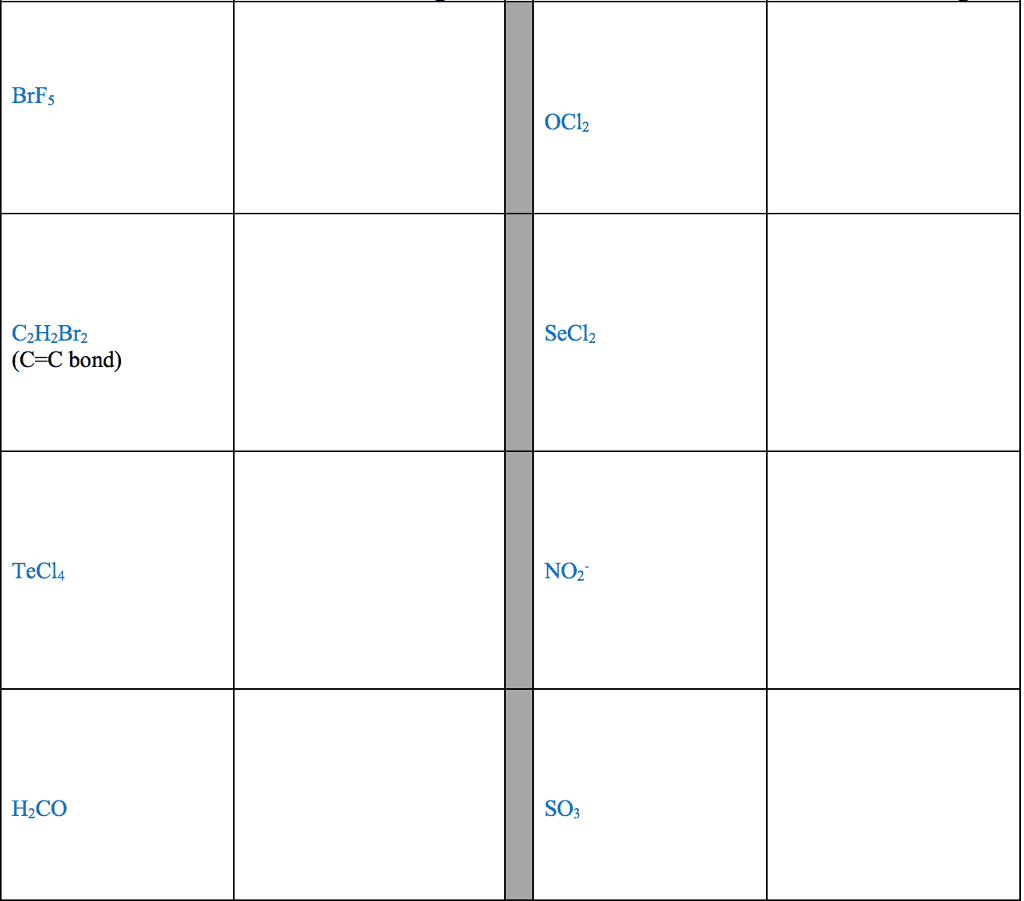
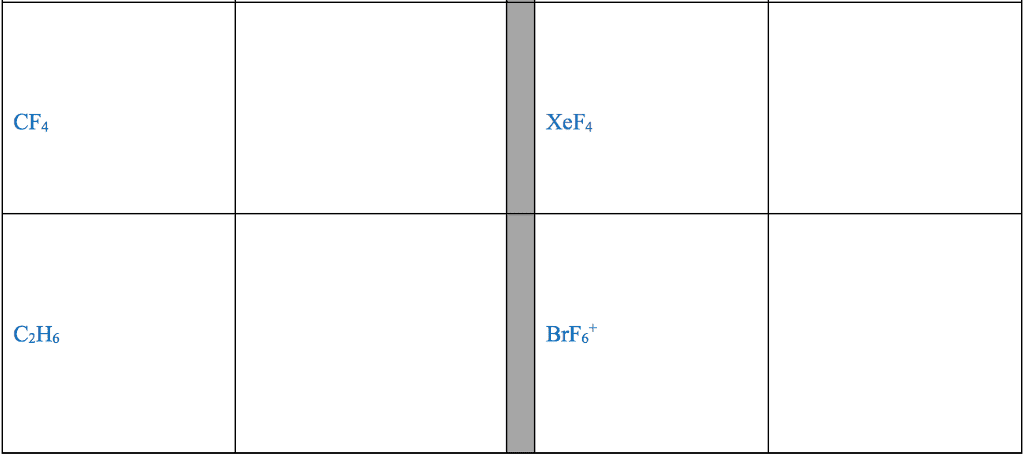
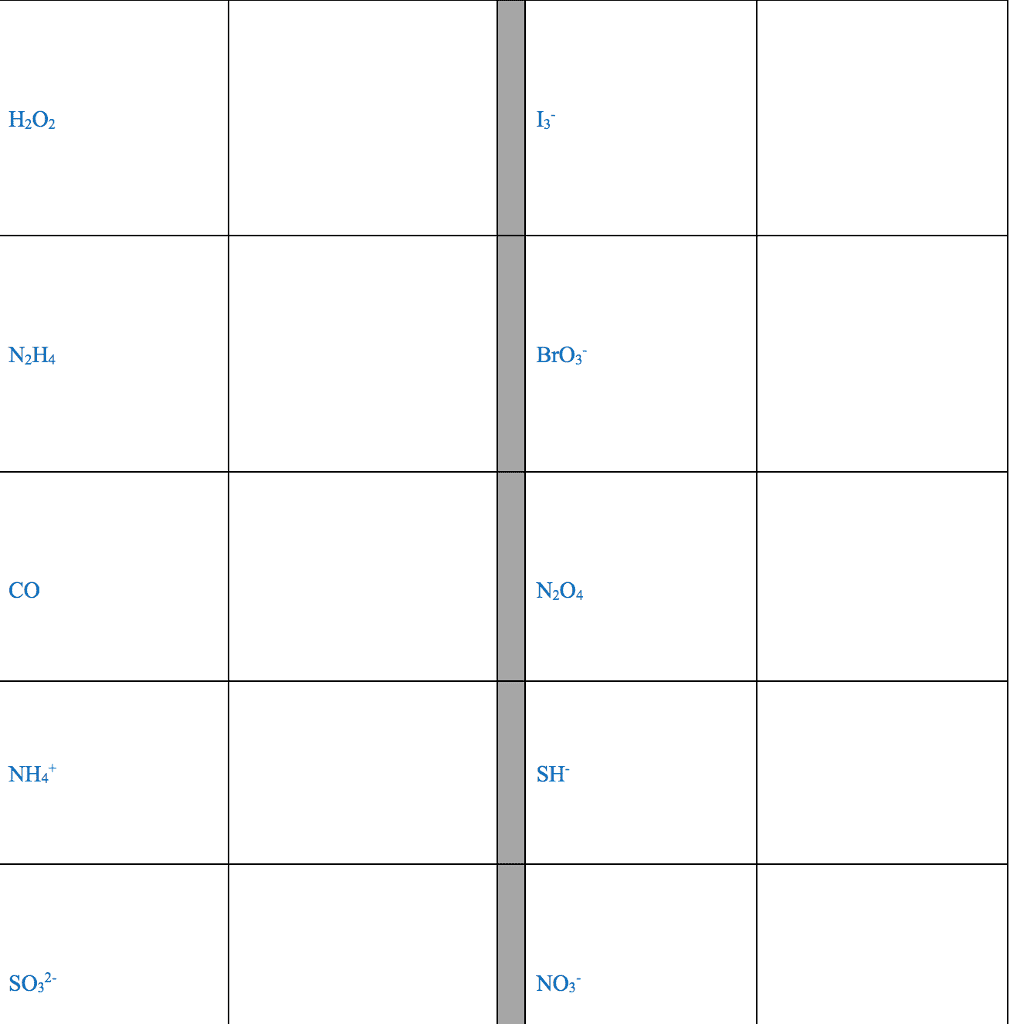
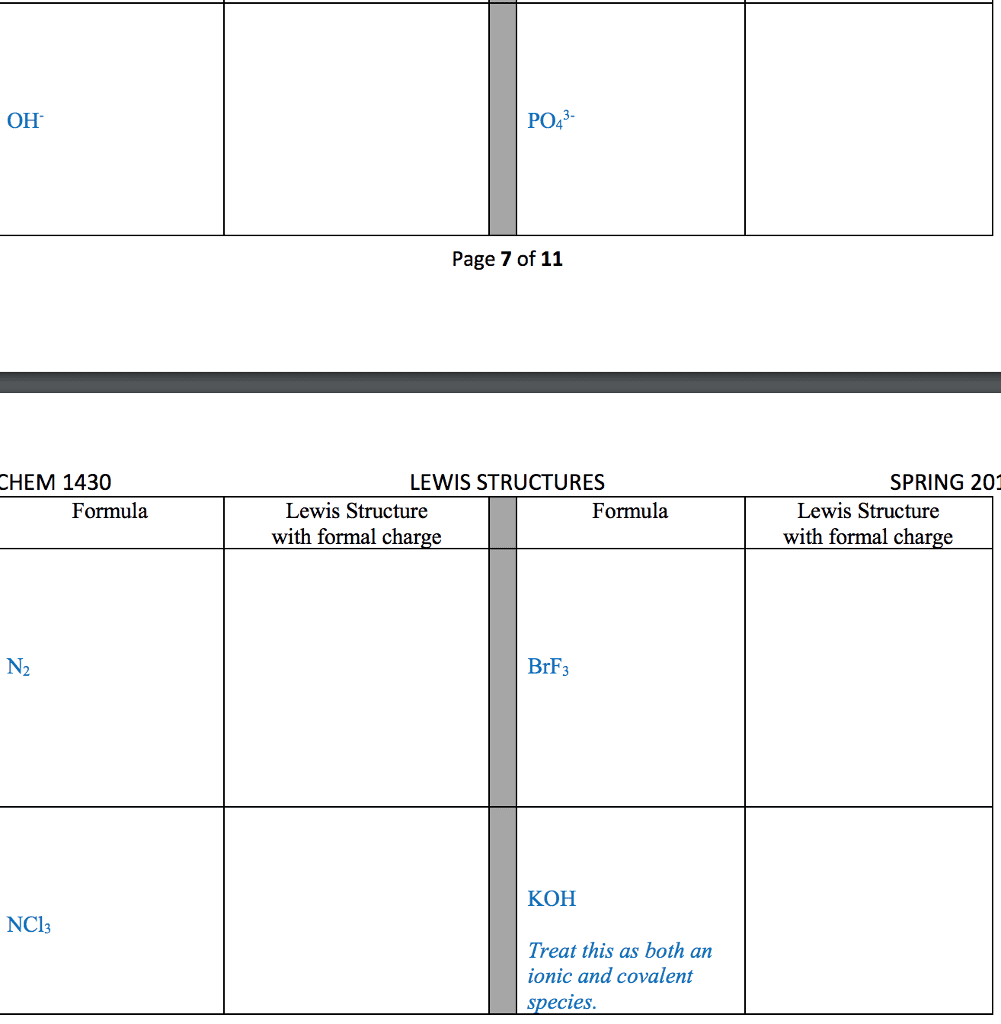
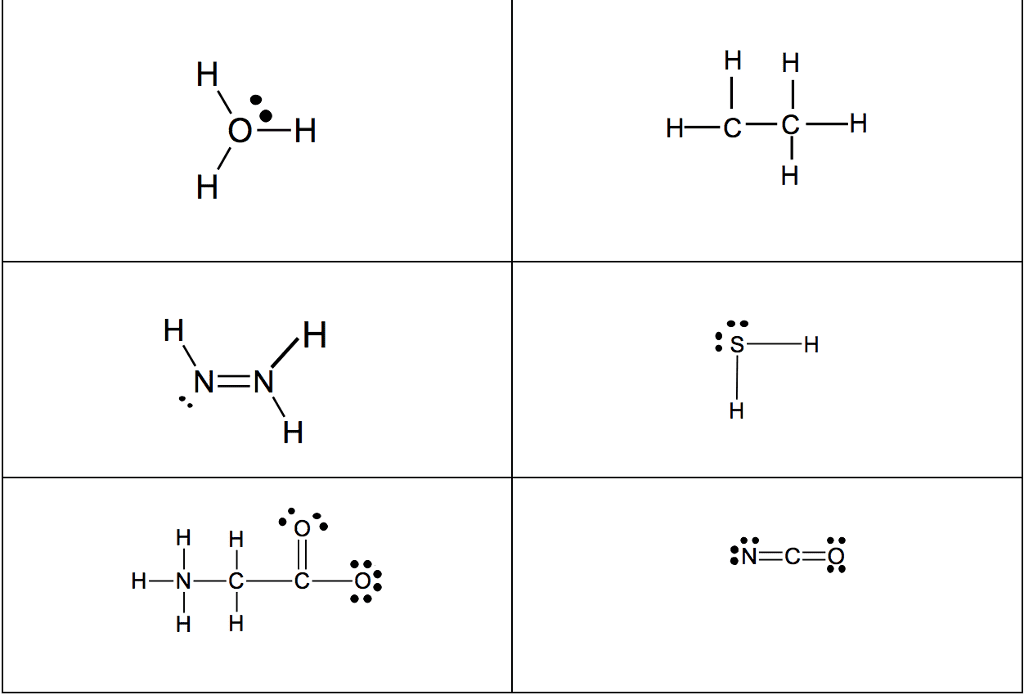
28th feb 2018: lecture three atoms and molecules: noble gas electron number: The valence shells of noble gases cannot accept additional electrons, as their shells are already full. Valence electrons drive chemical reactions and facilitate chemical bonding, as atoms become more stable by losing or sharing their valence electrons. Octet rule: main-group elements tend to combine in such a way that each atom has eight electrons in its valence shell, giving it the same electron configuration as a noble gas. When metals and non-metals react, electrons tend to be gained or lost the species is able to achieve noble gas electron number. (the number of protons does not change its identity when electrons are gained or lost: lewis dot structure: A pictorial way to represent the valence electrons of atoms and molecules, which are known as lewis dot diagrams. It is useful for representing changes, when valence electrons are added or removed from an atom or molecule: bonding lewis model: