CHEM1831 Lecture Notes - Lecture 2: Gastrointestinal Bleeding, Mass Spectrometry, Radiocarbon Dating
Document Summary
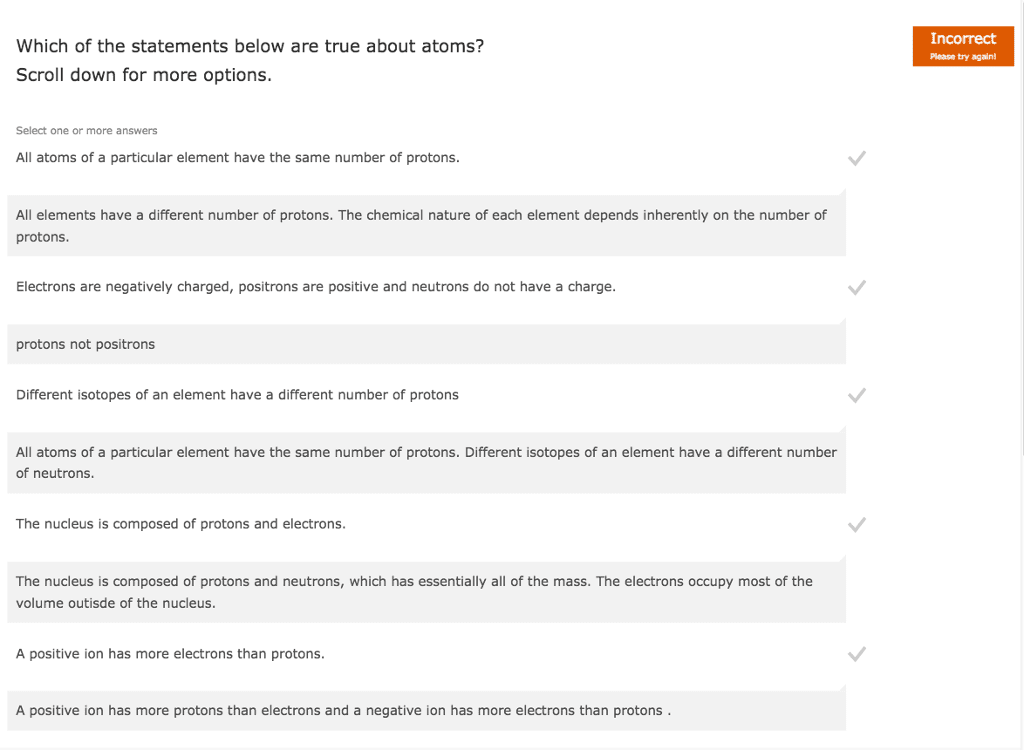
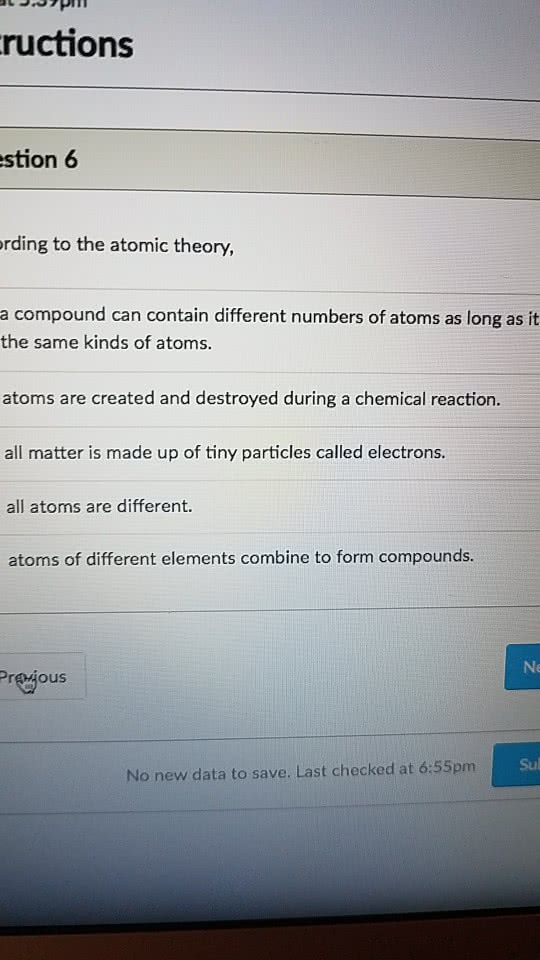
All atoms of any element are the same. Atoms of different elements can combine to form compounds. In any compound the numbers and kinds of atoms remain the same. Compound-pure substance made of 2 or more chemically bonded atoms of different elements. Molecule - stable entity of 2 or more chemically bonded atoms - may be the same element. Molecular formula - number of atoms of each element per molecule. Atoms: made up of 3 subatomic particles electrons, protons and neutrons. All atoms have the same number of protons in the nucleus and electrons in surrounding shells. Isotopes same element, different mass e. g carbon 12 and carbon 13. All atoms of the same element have the same number of protons isotopes. Radiocarbon dating t1/2 =5730 years therefore doesnt work with dinosaurs. E. g chromium-51 monitoring blood cells, diagnosing gastrointestinal bleeding. Tc-99m is the most widely used medical isotope in the world. Neutral atoms number of protons = number of electrons.