CHEM1011 Lecture Notes - Lecture 22: Intermolecular Force, Phase Transition, Heat Capacity

Lecture 22: Phase eatios + Hess’s La
Recap: latent heats
- Energy absorbed by chemical systems increases molecular motions and can break bonds
or intermolecular forces
- Heat capacity quantifies how energy is absorbed within a single phase (solid, liquid, gas).
Latent heats quantify how energy is absorbed in a phase transition
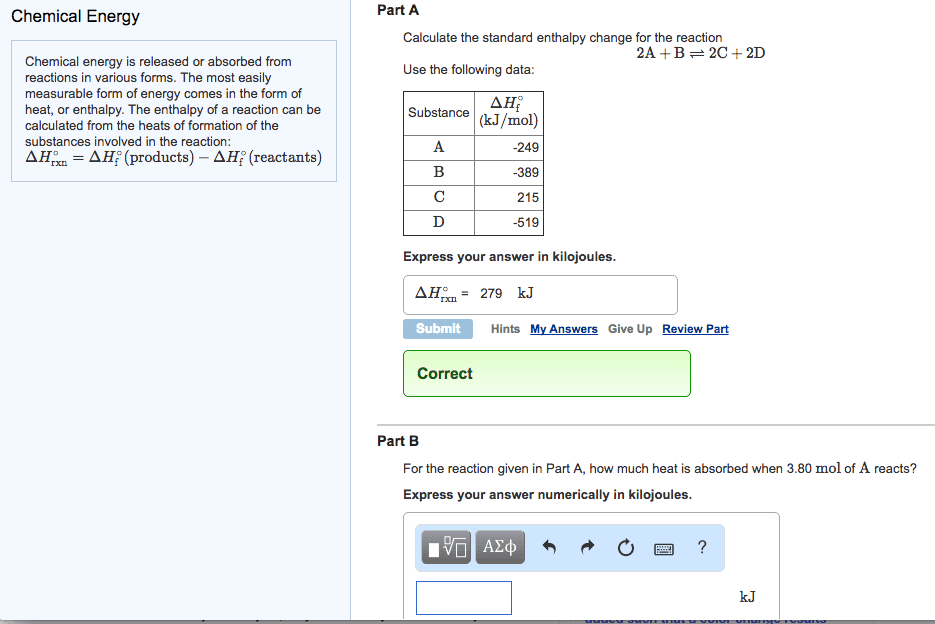
Phase reactions
The latent heats (enthalpies) of a reverse process is simply the negative of the forward process
Reaction diagrams: representing enthalpy change
In a chemical reaction, we represent the change in enthalpy as the difference between the
enthalpies of the products and the reactants. e.g. we can represent the heat of the reaction
The enthalpy of the products is less than that of the reactants. Hence the enthalpy change is
negative. That is, the reaction is exothermic, and heat is released into the surroundings. For this
reason, exothermic reactions may be written with heat as a product:
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Energy absorbed by chemical systems increases molecular motions and can break bonds or intermolecular forces. Heat capacity quantifies how energy is absorbed within a single phase (solid, liquid, gas). Latent heats quantify how energy is absorbed in a phase transition. The latent heats (enthalpies) of a reverse process is simply the negative of the forward process. In a chemical reaction, we represent the change in enthalpy as the difference between the enthalpies of the products and the reactants. e. g. we can represent the heat of the reaction. The enthalpy of the products is less than that of the reactants. That is, the reaction is exothermic, and heat is released into the surroundings. For this reason, exothermic reactions may be written with heat as a product: Our reaction diagram tells us immediately that the reverse reaction. Is endothermic, and its enthalpy change is the negative of. P, v, t, e and h (and many other quantities) are state functions.