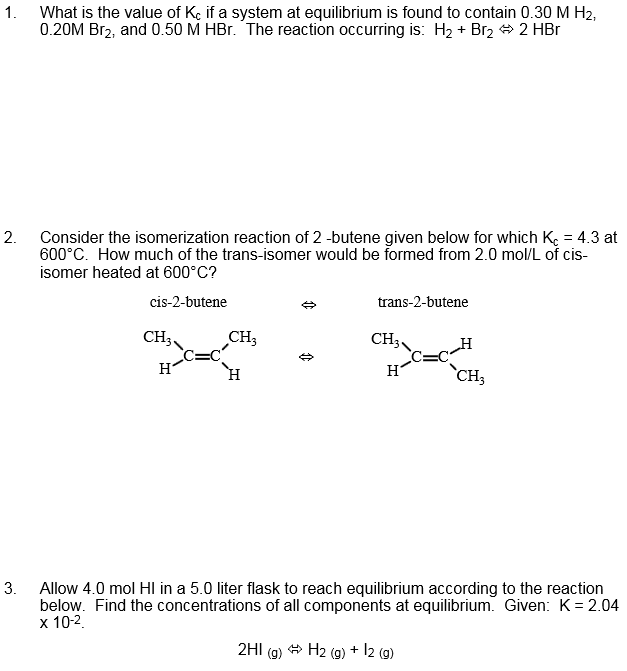
CHEM1011 Lecture Notes - Lecture 26: Butene, Equilibrium Constant, Reaction Quotient
24 views4 pages
9 Jun 2018
School
Department
Course
Professor

Lecture 26: Equilibrium calculations
Solving for equilibrium concentrations- 1
If we know the initial conditions for a reaction and the equilibrium constant, then it is generally
possible to solve for the equilibrium compositions of all products and reactants in a chemical
system
The simplest example is an isomerization reaction, where a single reaction changes its structure
to form the product. If the initial concentration of cis-2 butene is 0.01 M and no trans-2 butene
is present
These initial concentrations describe the system before equilibrium is established. As time
passes, these concentrations change until equilibrium is achieved.
Solving for equilibrium concentrations- 2
find more resources at oneclass.com
find more resources at oneclass.com
Unlock document
This preview shows page 1 of the document.
Unlock all 4 pages and 3 million more documents.
Already have an account? Log in
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232