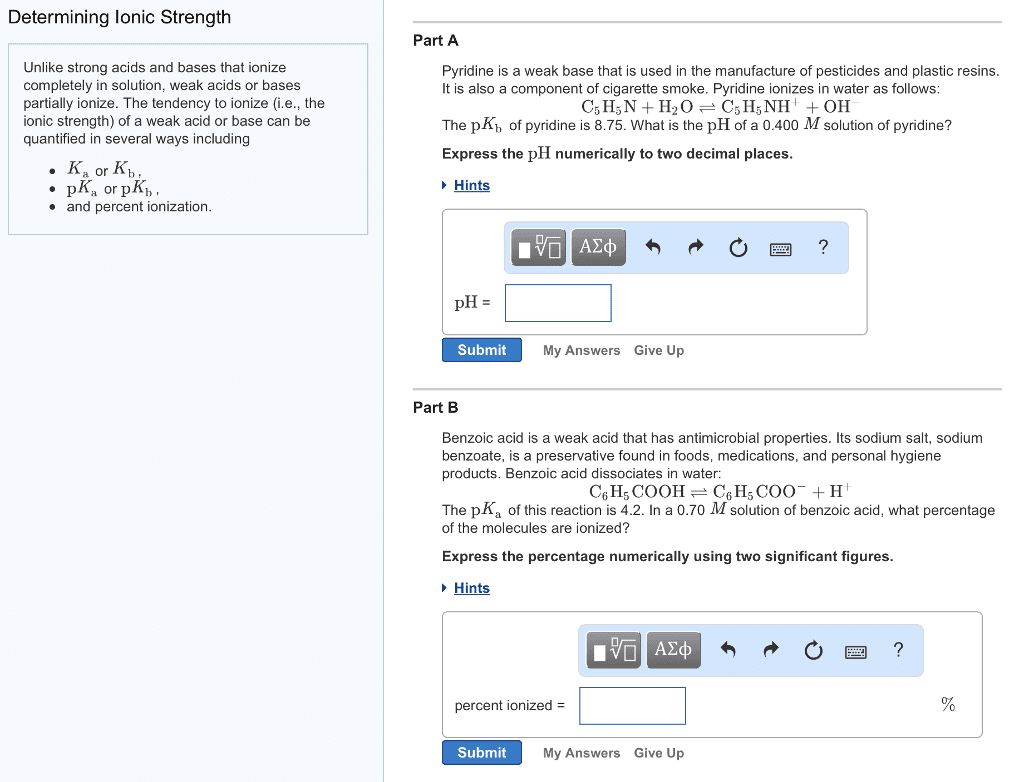
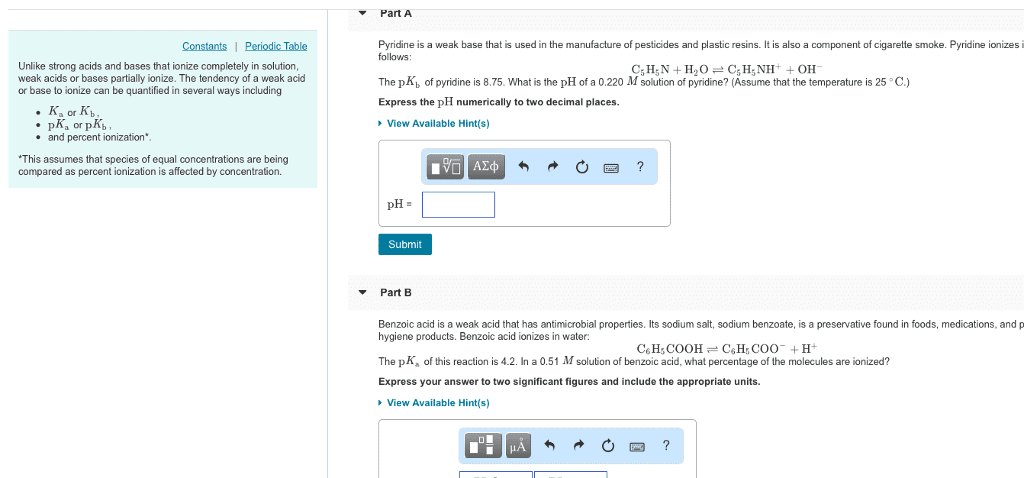
CHEM1011 Lecture Notes - Lecture 28: Equilibrium Constant, Acid Dissociation Constant, Weak Base

Lecture 28: weak acids and bases
Weak acids and equilibrium
- A strong acid has equilibrium to the right
- A weak acid has equilibrium to the left
Equilibrium equation:
Ka is the acid ionisation constant
Note: the concentration of water is assumed to be constant
Weak acids
Most acids or bases are weak
- They do not completely ionise in water
The larger the value of Ka the stronger the acid and the lower the value of pKa.
HA= acid
find more resources at oneclass.com
find more resources at oneclass.com

2
Relationship between Ka and pKa
E.g. 1
• Fid the pH of 0.1 M acetic acid (CH3COOH (HAc))
find more resources at oneclass.com
find more resources at oneclass.com