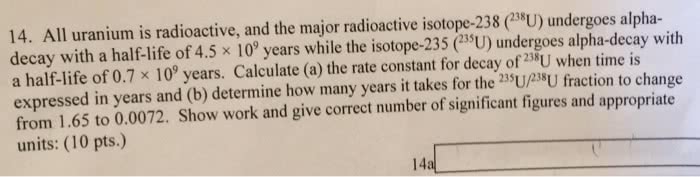CHEM1111 Lecture Notes - Lecture 3: Exponential Decay, Gamma Ray, Specific Activity
Document Summary
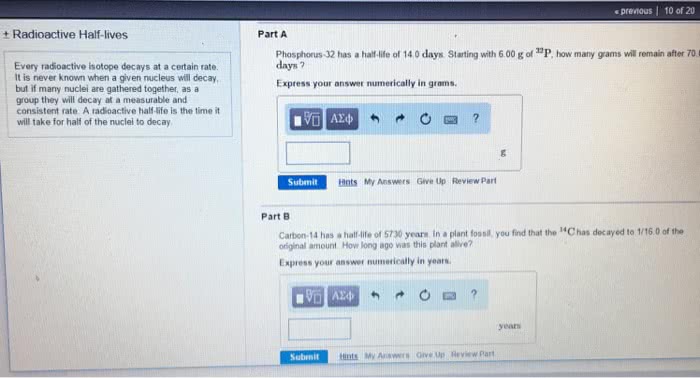
Radioactive nuclei undergo spontaneous decay to become a stable nucleus. All radioactive nuclides have different rates and modes of decay (i. e. alpha, beta, gamm. Half life: the time required for half of the nuclei in a sample to undergo a decay event. When we plot this into a curve, it is an exponential decay curve. The decay constant (k) is characteristic of the particular radioactive nuclide, and does n amount of sample. Rate of decay depends on the number of nuclei present. Formula: a= - (change in number of nuclei) / (change in time) Important: put the minus (-) in front when rate is negative because there is no su decay amma decay) ent oes not depend on the no such thing as negative. To find the half life, substitute t= t1/2 and n= n0/2. Activity (a): the number of nuclei that disintegrate per second. Specific activity is the activity per gram of radioactive nuclide.