CHEM1112 Lecture Notes - Lecture 4: Phenols, Racemic Mixture, Halocarbon

• Identify and name simple alcohols and phenols
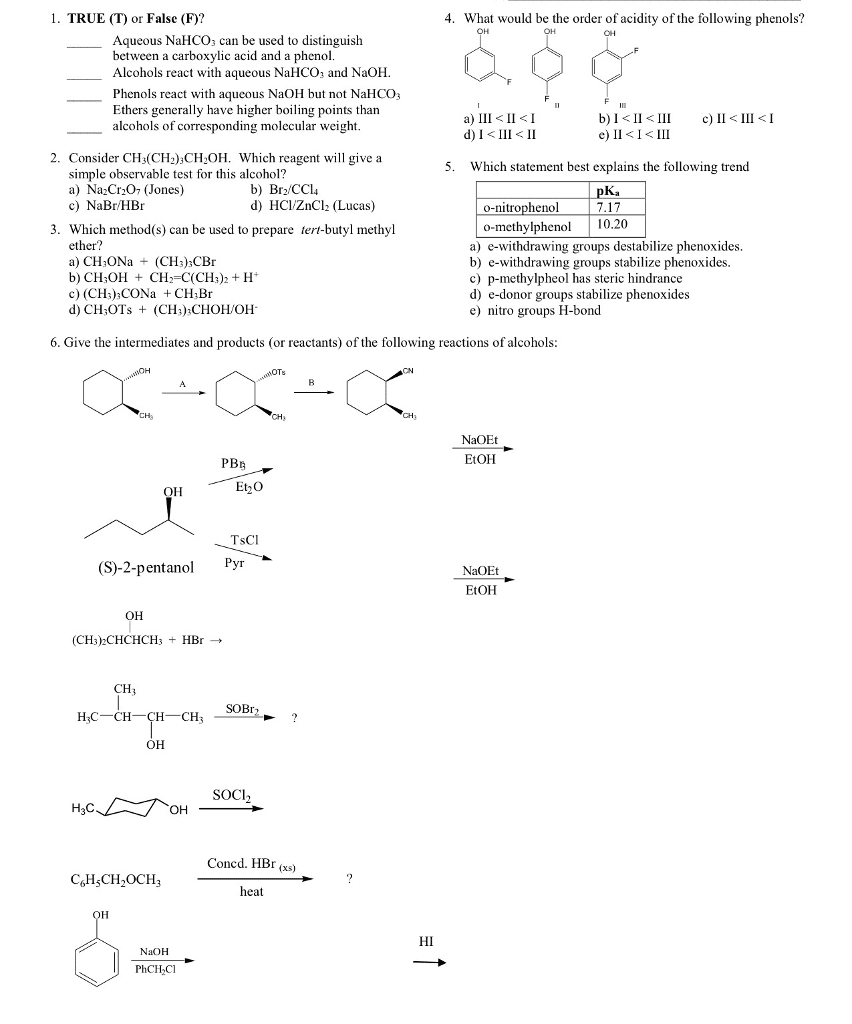
• Explain why phenols are more acidic than alcohols
find more resources at oneclass.com
find more resources at oneclass.com

Alcohols: electronegative OH, polar, H-bonding, high BPs, soluble in water.
Grignard reagents reacted with aldehydes or ketones, intermediate salts hydrolyzed to alcohols.
Nucleophillic Grignard reagent attacks partially positive C of carbonyl group.
Least acidic to most acidic:
Alcohols < water < thiols and Phenols.
In the phenoxide anion, negative charge not localised on the O atom (as it is in hydroxides). Negative
charge is delocalised about the aromatic ring, delocalisation of electrons/charge= greater stability.
• Predict the products of oxidation of an alcohol and give the reagents required to form this
reaction
This is substitution- Nucleophilic- second degree (SN2). Reaction rate proportional to concentrations of
both nucleophile and electrophile. Intermediate: highest energy molecule, where 3 substituents in place
move to trigonal planar arrangement, partial breaking of C-X bond, then full broken C-X and fully formed
C-Nu in the final product.
When reaction rate proportional to concentrations of the organohalide only: Substitution-nucleophilic-
first order (SN1)- 2 step process for tertiary that does’t react through SN reactio. SN: slow step
waits for C-Br to step, unimolecular step to take place, wait for C-X to break from a single molecule,
unlike SN2 reactions that need the molecules to interact for reaction to begin.
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Identify and name simple alcohols and phenols: explain why phenols are more acidic than alcohols. Alcohols: electronegative oh, polar, h-bonding, high bps, soluble in water. Grignard reagents reacted with aldehydes or ketones, intermediate salts hydrolyzed to alcohols. Nucleophillic grignard reagent attacks partially positive c of carbonyl group. In the phenoxide anion, negative charge not localised on the o atom (as it is in hydroxides). Negative charge is delocalised about the aromatic ring, delocalisation of electrons/charge= greater stability: predict the products of oxidation of an alcohol and give the reagents required to form this reaction. Reaction rate proportional to concentrations of both nucleophile and electrophile. Intermediate: highest energy molecule, where 3 substituents in place move to trigonal planar arrangement, partial breaking of c-x bond, then full broken c-x and fully formed. When reaction rate proportional to concentrations of the organohalide only: substitution-nucleophilic- first order (sn1)- 2 step process for tertiary that does(cid:374)"t react through sn(cid:1006) reactio(cid:374).