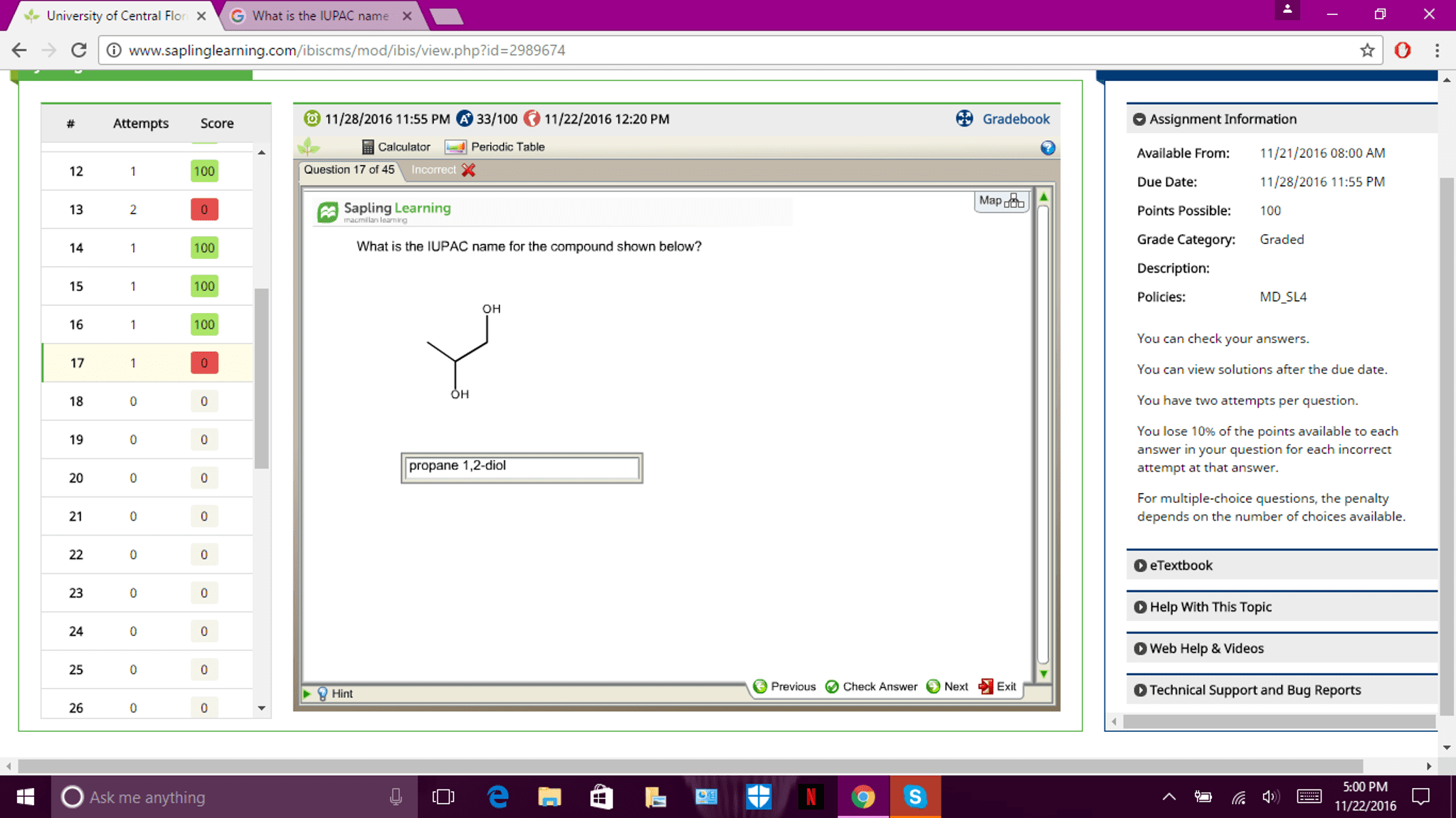
CHEM1112 Lecture Notes - Lecture 7: Ketone, Elimination Reaction, Primary Alcohol
Identify and name simple alcohols and phenols
7.1 (wk 5)
Monday, 28 August 2017
10:20 PM
7. Alcohols Page 1
Explain why phenols are more acidic than alcohols
7.2
Monday, 28 August 2017
10:21 PM
7. Alcohols Page 2
Document Summary
Identify and name simple alcohols and phenols: alcohols page 1. Explain why phenols are more acidic than alcohols: alcohols page 2. Predict the products of oxidation of an alcohol and give the reagents required to perform this reaction. Alcohols have high bps - many soluble in water. Second one has no reaction because no hs to remove on carbon which oh is attached to. For oxidation must have alpha hs (ie. hs attached to alpha carbon, which is carbon attached to oh group) In this mechanism, you lose one alpha h and increase o content. Note: you cannot oxidise a tertiary alcohol (no h attached to alpha carbon for oxygen to remove) Biological example of oxidation of alcohol: alcohols page 4. Screen clipping taken: 2/10/2017 2:23 pm: alcohols page 5. Predict the products of the elimination of alcohols, using zaitsev"s rule, and give the reagents required to perform this reaction. Protonation is fast, loss of water is slow.