CHM1011 Lecture Notes - Lead, Chromate And Dichromate
Document Summary
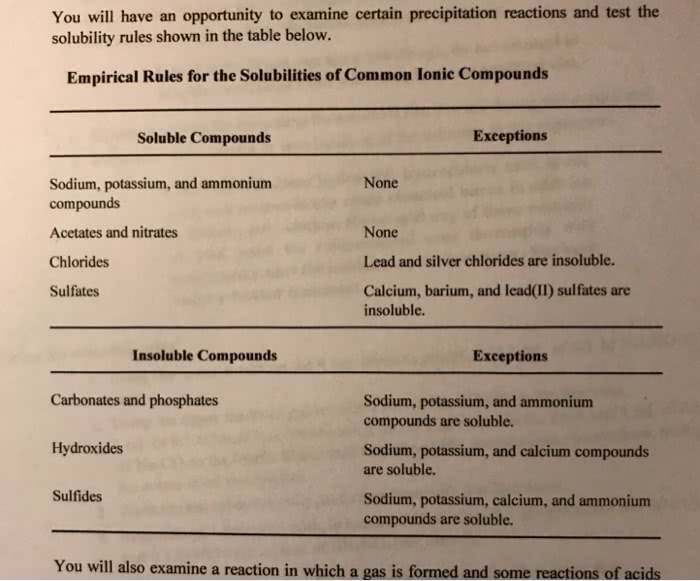
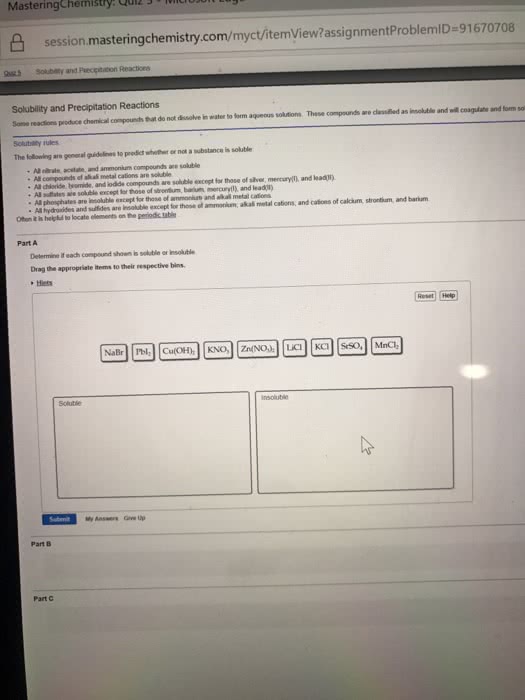
All salts containing sodium (na+), potassium (k+) or ammonium (nh4 soluble. All salts containing chloride (cl-), bromide (br-) or iodide (i-) are soluble except salts formed with silver (ag+), and lead (pb2+). 2+), lead (pb2+), calcium (ca2+), strontium (sr2+) or barium (ba2+). All salts containing phosphates (po4 except compounds formed with sodium (na+), potassium (k+) or ammonium (nh4. All salts containing hydroxide (oh ) are insoluble except compounds formed with sodium (na+), potassium (k+), ammonium (nh4. Hydroxide salts containing strontium (sr2+) or calcium (ca2+) are borderline soluble. All salts containing sulfide (s2 ) are insoluble except compounds formed with sodium (na+), potassium (k+), ammonium (nh4 strontium (sr2+) or barium (ba2+). Here are some ions which might not be in chapter 3 of the text by blackman et al.