CHEM1011 Lecture Notes - Lecture 7: Valence Electron, Ionic Bonding, Octet Rule
Document Summary
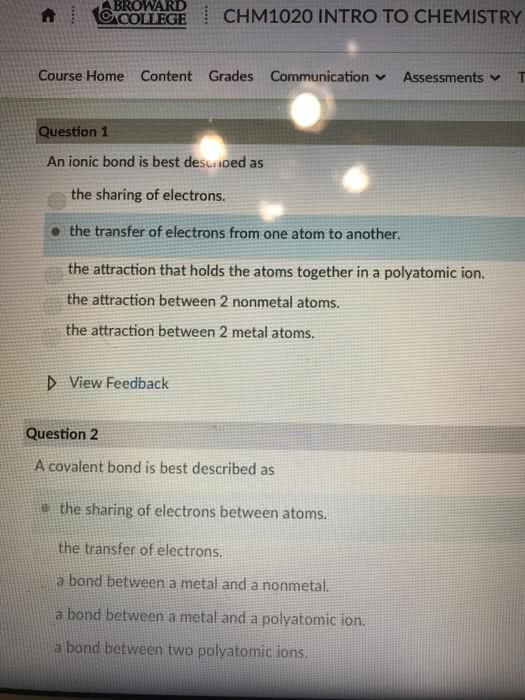
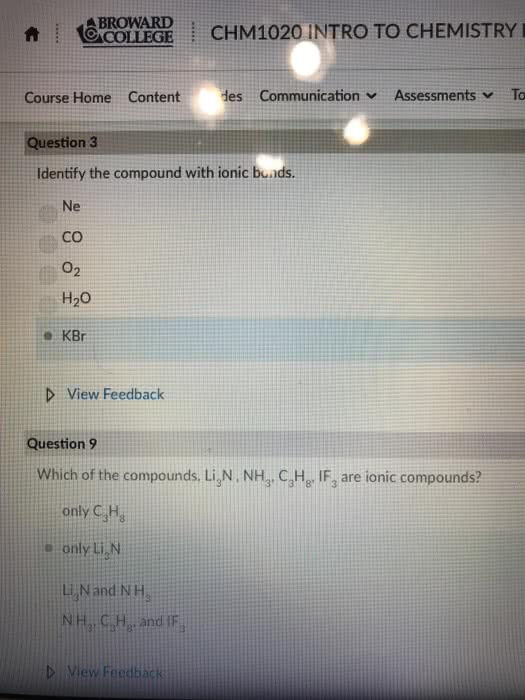
Ionic bonds form when a metal reacts with a non metal. An ionic bond is the electrostatic attraction between oppositely charged ions in a crystal structure. In reactions, we simplify our representations of ionic compounds by considering only the smallest number of atoms required to form a compound with a neutral charge (empirical formula), e. g. nacl. But ionic compounds actually form crystal structures where each ion is surrounded by several other ions of the opposite charge to maximise the electrostatic interaction. A covalent bond is formed when electrons are shared between two atoms. The electron sharing can be even or uneven. Covalent bonds normally form so that each non hydrogen atom has 8 electrons around it: the octet rule . It takes two electrons to make a single covalent bond, one for each atom. Transition metals are a different story all together. They don"t tend to obey the octet rule. Valence electrons are not shared exclusively between two atoms.