CHEM 1001 Lecture Notes - Lecture 1: Steric Number, Lattice Energy, Trigonal Pyramidal Molecular Geometry

7
CHEM 1001 Full Course Notes
Verified Note
7 documents
Document Summary
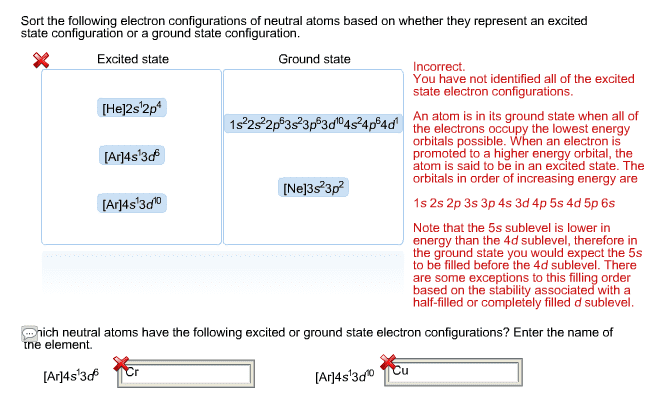
Answer: the element with more unpaired electrons will have the higher spin. Mo has ground-state configuration [kr] 5s1 4d5; that of tc is [kr] 5s2 4d5: Answer: write configurations using the standard filling procedure, but watch for exceptions. (a) n: z = 7, [he] 2s2 2p3 (b) ti: z = 22, [ar] 4s2 3d2 (c) as: z = 33, [ar] 4s2 3d10 4p3. 4. one element has these ionization energies and electron affinity(all in kj/mol) : ie1 =376 , ie2 = (d) xe: z = 54, [xe] or [kr] 5s2 4d10 5p6. The gain in lattice energy for this compound more than offsets the additional energy required to form the ions, but the energy required to remove the third electron from ba is prohibitive. Answer: the value of ie2 is almost 10 times that of ie1, indicating that the second electron removed is a core electron rather than a valence electron.