CHEM 1002 Lecture Notes - Lecture 1: Tasmania, Joule, Diethyl Ether
51 views5 pages
26 Jan 2015
School
Department
Course
Professor
Document Summary
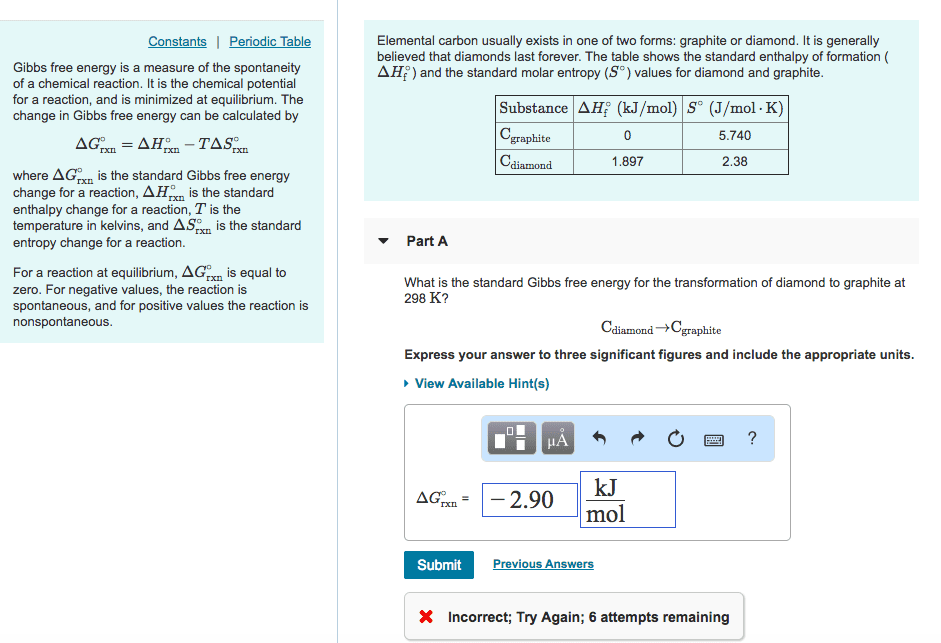
Thus, when a change occurs, the change in entropy (d s) may be either positive or negative. A positive d s is a favorable change, i. e. one which tends to make the change spontaneous. Recall that a negative d h (i. e. heat released, an exothermic reaction) is also a favorable situation, Recall that a negative d h (i. e. heat released, an exothermic reaction) is also a favorable situation, i. e. one which tends to make the reaction spontaneous. Thus, a reaction that has both a negative. D h and a positive d s will be spontaneous no matter what the temperature is. In order to quantify these effects so as to be able to predict whether or not a reaction will be spontaneous, we need to use the change in free energy, d g, for the reaction. Co2(g) + 2 h2o ch4(g) + 3 o2(g)
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232