CHEM 1101 Lecture Notes - Lecture 9: Lincoln Near-Earth Asteroid Research, Bond Order, Lone Pair
Document Summary
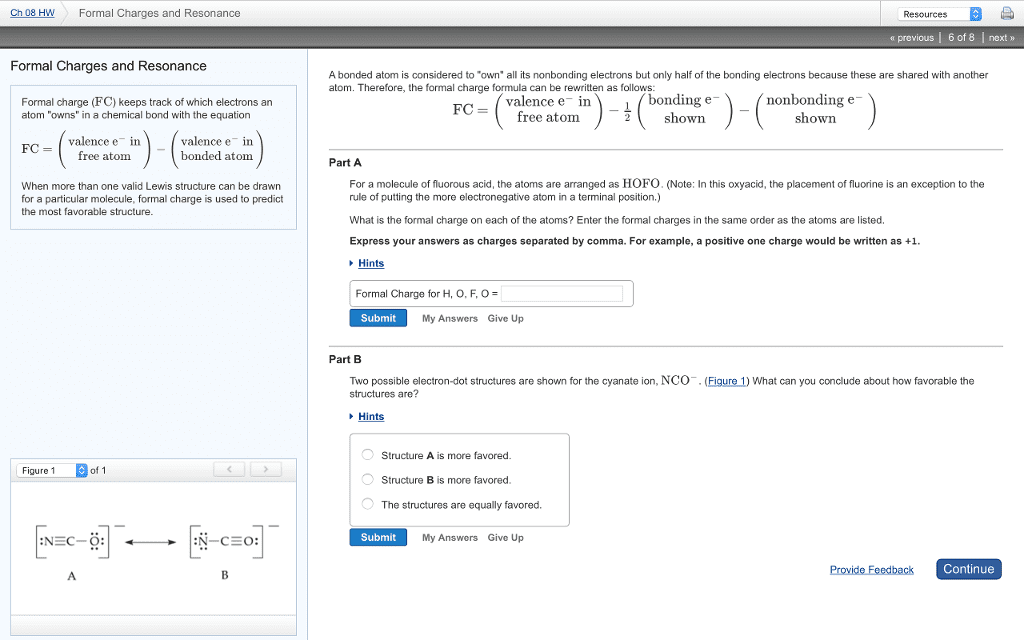
If: central atom is row 3 or lower. And: at least one positive and one negative f. c. Move a lone pair of electrons off the element with the negative. Formal charge and into the bond joining it to the element with the. When a lewis structure has fewer multiple bonds than there were equivalent positions where you could have placed the multiple bond(s), the multiple bonds are shared among. Draw enough resonance structures to show every possible position having every possible bond order. It only takes two resonance structures to show that so 4 among 4 equivalent s to o bonds. (seen below) No resonance = simply count the lines and call the bonds what they are. If resonance = average bond order (see right side of picture) All the electrons in one individual chemical bond (2 in a single bond, 4 in a double bond, 6 in triple bond) have to stay in the same region of space.