BIOC 2300 Lecture Notes - Lecture 3: Surface Charge, Stage Name, Acid Dissociation Constant
Document Summary
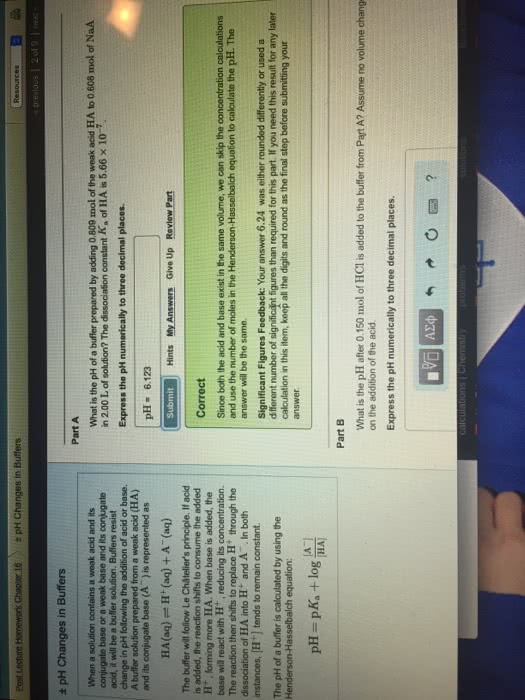
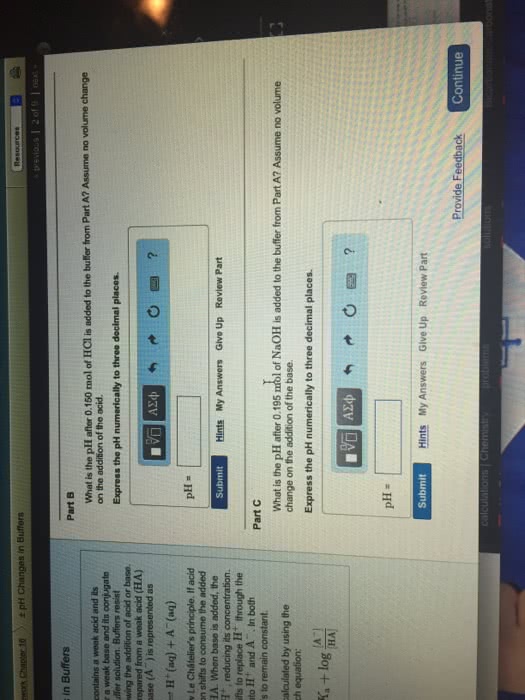
Biochem lecture 3: acid-base chemistry and buffers. Objectives: review the fundamentals of acids and bases. Acid, base, conjugate acid/base pair, kw, ka, ph, pka. As you change the ph of ubiquitone, the surface charge changes completely. As we go towards basic ph, the charge becomes negative. This is why ph matters and the ability of different chemcials to occur. Hydronium ions are very mobile due to h bonding network. The aquesuous solutions the general definition of acids/bases are from the bronstead. Ocygen is electronegative and withdraw some of the electric charge. Self- dissociation/autoionization = same thing, where water is able to split apart, to some degree, to hydronium ions and hydroxide ions. Free protons don"t really exist but they exist as hydronium ions. Whats special about hydronium is that it diffuses readily due to the flickering clusters in solution make and break bonds readily.