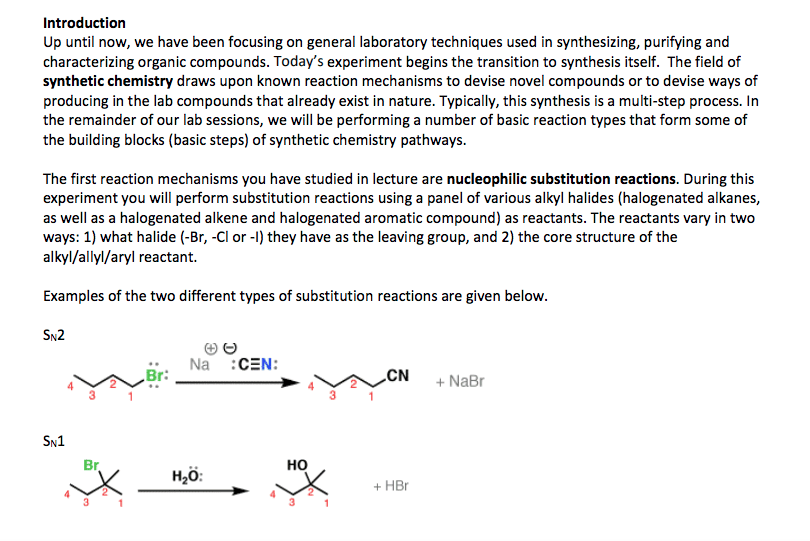
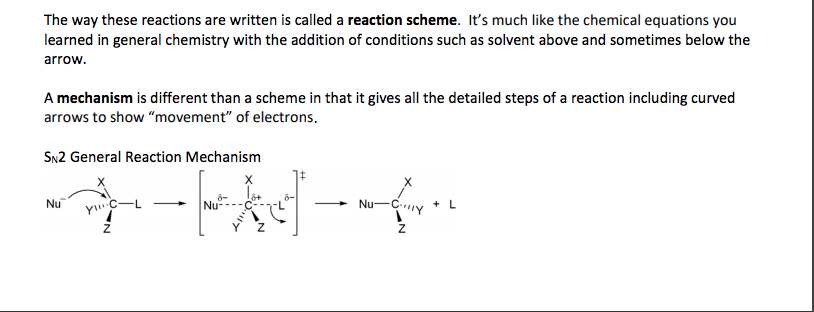
CHY 103 Lecture Notes - Lecture 6: Calcium Carbide, Joule
Document Summary
We have seen that calorimetry can be used to determine the enthalpy of reaction. Example: the one-step combustion of carbon to form carbon monoxide can be represented by. C (s) + 2 o2 (g) co (g) Rh = -110. 5 kj mol-1 or by the two-step process, C (s) + o2 (g) co2 (g) Co2 (g) co (g) + 2 o2 (g) The advantage: we can calculate the rh for almost any process by combining a series of reactions of known rh. Example: calculate rh for the reaction given the following information: No (g) + o (g) no2 (g) (1) (2) o3 (g) 3/2 o2 (g) (3) o2 (g) 2 o (g) No (g) + o3 (g) no2 (g) + o2 (g) Since enthalpies depend on conditions such as temperature and pressure a common set of conditions, the standard state was defined in order that enthalpies could be compared.