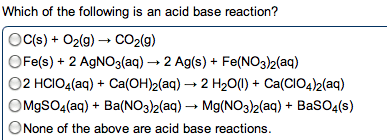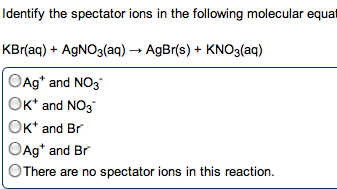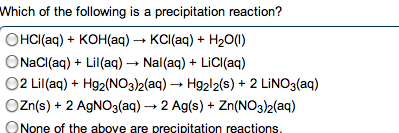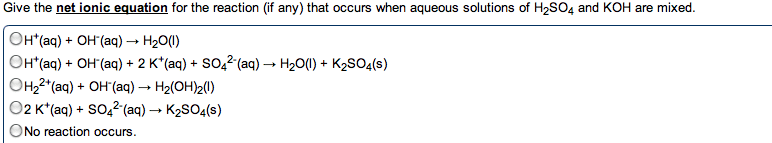CHEM 1040 Lecture Notes - Lecture 24: Sodium Acetate, Conjugate Acid, Chemical Equation
93 views3 pages
7 Nov 2017
School
Department
Course
Professor
Document Summary
Chem 1040- lecture 24- k values , ph and buffers. You need to know two things for aqueous buffer systems: Did a chemical reaction occur: determines from net ionic equation. Is the process complete as written: determined from k value. If the reaction involves a weak acid/base, a reaction occurs. If the reaction involves a strong acid/base, the compounds only dissociated. K+, na+, li+, ca2+, ba2+, sr2+ from oh- We want the k value for the reaction going from left to right. The k value is determined from the species that is mixing with the water. When water is on the left, a dissociation or normal process occurs: the k value is used directly. Ex: show the net ionic equations and k expressions for the following: sodium acetate in water. Kch3coo- = kw / kch3cooh: acetic acid mixed with sodium hydroxide. Ch3cooh + oh- --> h2o + c h3coo- This is the reciprocal of the above reaction, so:
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232