CHEM 123 Lecture 2: CHEM 123 LEC 2 UBC CHEMISTRY THERMODYNAMICS WINTER 2019

48
CHEM 123 Full Course Notes
Verified Note
48 documents
Document Summary
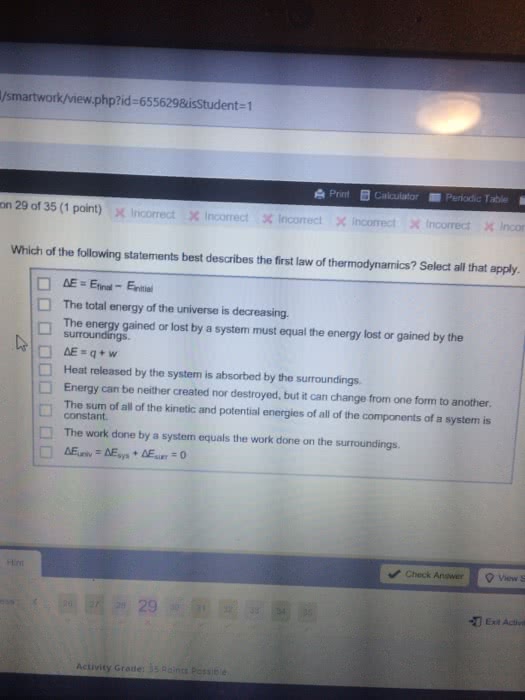
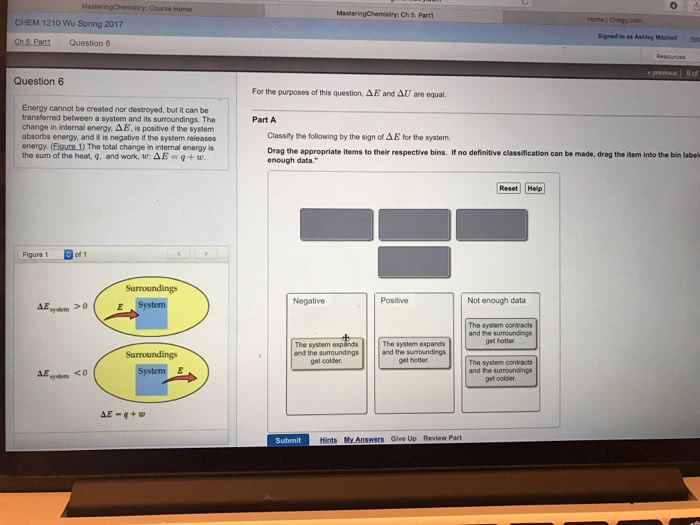
The first law of thermodynamics: conservation of energy: energy can neither be created nor destroyed, but it can be converted from one form to another. There exists a state variable called internal energy e. E is the sum of all forms of energy in the system. Change in energy (e) = q + w (for all paths) Internal energy: e[j], an extensive property: for any kind of energy. Change in energy = ef ei: equation of state for ideal gas, ideal gas isotherm. Internal energy (e): all forms of energy within a system, it includes. P1v1=p2v2: movement of molecules, translational, rotational, movement, energy within the molecule, vibrational, bonds, energy associated with the interactions between molecules, potential energy \ Internal energy of ideal gas: e of the ideal gas (pv=nrt) is a fxn of temperature only. For an ideal gas, if delta t=0, then delta e=0: for monatomic ideal gas,