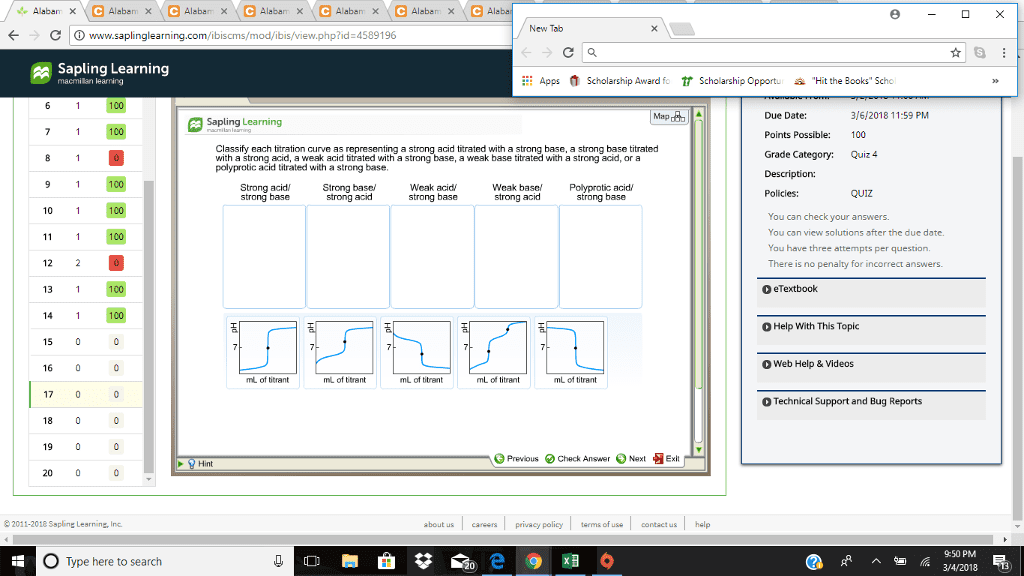
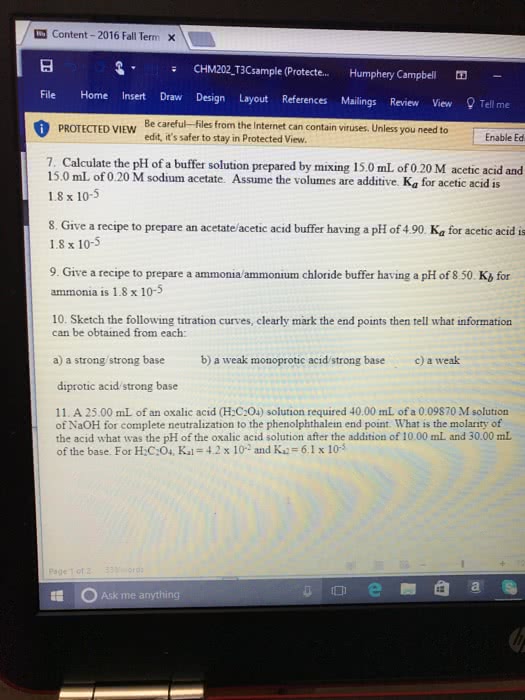
CHM110H5 Lecture Notes - Lecture 1: Simultaneous Equations, California State Route 1, Hydronium
Document Summary
Purpose: this experiment was to determine the heat of ionization of a weak acid by calorimetry using a strong acid and a strong base, and a weak acid and a strong base. Heat capacity of calorimeter s e e r g e d e r u t a r e p m e. 0 s e e r g e d e r u t a r e p m e. Neutralization of weak acid s e e r g e d e r u t a r e p m e. 1. |heat lost by hot h20|= |heat gained by cold h20+ heat gained by calorimeter| = |50 g * 4. 184* -8. 4|= |50 * 4. 184* 6. 4| + |cp * 6. 4| Cp= 418. 4j/6. 4= 65. 38j/k-1: mol base added= mol h30 neutralized. =0. 0503mol heat released by reaction= heat gained by a+b +calorimeter. H30 +oh 2h20 (of 0. 0503 mol h30) q= -{( 50*4. 184*6. 44) +(50*4. 184*6. 44) + (65. 38 *6. 44)}