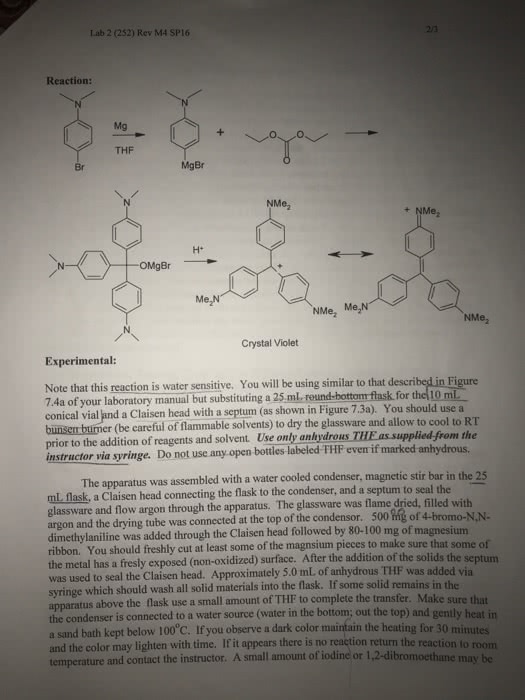
Please consider the preperation of benzoic acid: a Grignard Reaction.
Bromobenzene and diethyl ether is used in the formation of Grignard reagent.
PLEASE READ PROCEDURE TO HELP ANSWER THE QUESTION:
Procedure(question is in the pic below):
* add 1.2g of magnesium and 3 crystals of iodine to RB flask and immediately assemble the dry water condenser and drying tube containing cotton and anhydrous calcium chloride to RB flask
*then add 5.2 mL bromobenzene to 5 mL of diethyl ether to seperatory funnel and turn on condenser
* add about one third of ether-bromobenzene mixture down the condenser into the RB flask by momentarily removing drying tube. Place the drying tube back as quickly as possible.
* now add 30 mL of ether to the bromobenzene-ether mixture remaining in the seperatory funnel
*to initiate reaciton, place hands under flask and gently swril
* once the reaction maintains reflux, remove hands and set the flask down on a cork ring
*maintain the vigorousness of the reaciton by continuously adding 3.5 mL portions of diluted bromobenzene-ether mixture every 30 seconds
* make sure all of the bromobenzene-ether mixture has been added
* now obtain some dry ice, CO2 (s) and transfer 50 mL of this to the reaciton mixture inside the flask
*when all the dry ice has been added, add 80 mL of 6M HCl (aq) to flask
* finally, add 100mL of solvent grade diethyl ether (not anhydrous) and carefully stir
* transfer mixture to a 250mL seperatory funnel, allow layers to seperate and discard the lower aqueous layer
*now add 20 mL of 6 M NaOH (aq) and add 100mL of deionized water to seperatory funnel containing the ether solution
* drain the lower aqueous layer into a large beaker and slowly acidify using concentrated 12M HCl (aq). Stir the mixture and add HCl (aq) until pH is about 1-2 pH units.
*cool solution in ice-water bath and collect precipitate of benzoic acid
QUESTION:

Please do not forget to identify the phase that each compound is in. Thanks!
The overall reaction is given in the following pic:
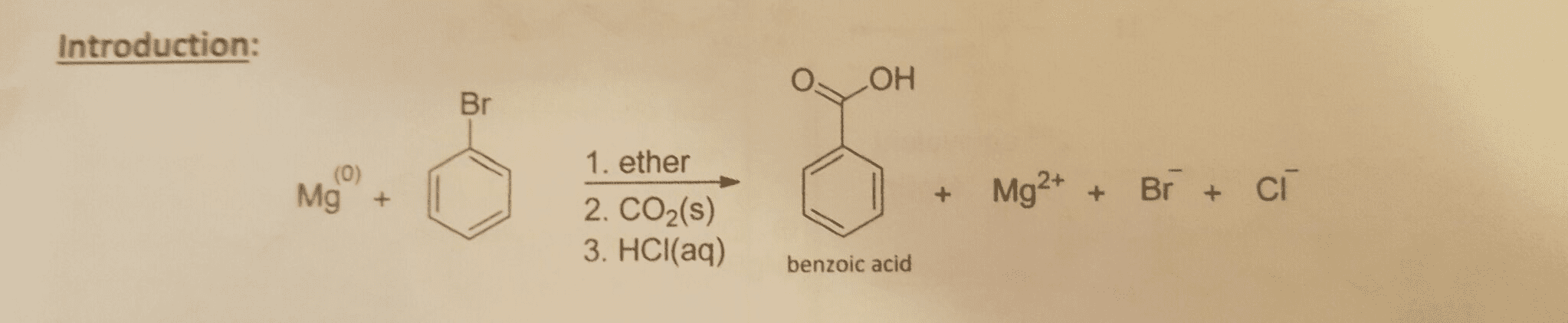
Questions (13.5 Marks Total): 1) In the procedure: following the addition of carbon d then 6 M Hcl(aq), diethyl ether was added and the layer discarded; then 6 M (aq) was added and the ether layer discarded Finally, concentrated HCl(aq) was added and a precipitate formed, which was filtered and cold deionized The purpose of these steps was to isolate, and thus purify, benzoic acid from H20. both water-soluble and organic-soluble impurities. (3 Marks Total) a) Provide a balanced equation for each step after the addition of Co2 b) State the impurities being removed in each step after the addition of CO2. In questions 1a and 1b; identify the phase (i.e. organic or aqueous) that each compound is in