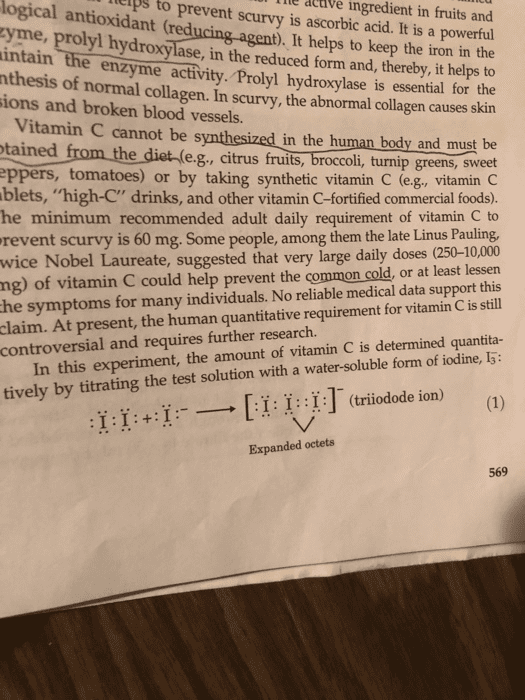47 EXPERIMENT 47 Report Sheet 1. The kind of fruit drink appl Jwic 2. Molarity of iodine solution ad 3. Titration results Sample 1 (100 mL) Sample 2 (20.0 mL) Sample 3 (30.0 mL) a. Initial buret reading b. Final buret reading c. Total volume of iodine ,',ã¼, mL u. 2 mL 2.5 mL 4.amL 10.5 mL 10.5 mL 20.5 mL solution used: (b)-(a) 4. The weight of vitamin C in the fruit drink sample: [(3c) x 1.76 mg/mL] 5. Concentration of vitamin C in the fruit drink (mg/100 mL): [(4)/volume of drink] x 100 O,20,30 5S 6. Average concentration of vitamin Cin the fruit drink 52 mg/100 mL
Post-Lab Questions 1. Use the average concentration of vitamin C from your data, and calculate the volume that would satisfy your minimum daily requirement of vitamin C. Show your work 2. Vitamin C is ascorbie acid. If you examine the structure of ascorbic acid given in the Background t. In the structure shown, the ascorbic acid is section (equation 2), there is no -COOH grou - as a lactone or cyclic ester. Write the equation for the hydrolysis (cleavage by addition of water) of E ste the lactone to the carboxylic acid. 3. Write the net ionic equation for the conversion of iodine, la, to iodide, I. What kind of reaction is this? 4. Refer to the Background section and examine the chemical reaction for the oxidation of ascorbic acid (equation 2) a. What happened to the hydrogen atoms that were present in the ascorbic acid? b. Acid was added in the titration, but it is not present in equation (2) in a stoichiometric amount. So, what is the purpose of the acid? 5. The iodine for this experiment is in the form of 15, and not 12 (see equation (1), Background section]. Why is this form of iodine used? 577
ts for General Organic and Blochemistry Bettelheim/Landesberg Vitamin C is oxidized by 12 (as 15) according to the following chemical reaction: HO-CH- CH2OH HO-CH-CH,OH O. . o 2HI н он он Oxidized product (MW 174) (MW 254) Vitamin C (MW 176) As vitamin C is oxidized by iodine, I2 becomes reduced to I. When the end point is reached (no vitamin C is left), the excess of 12 will react with a starch indicator to form a starch-iodine complex, which is blackish-blue in color. I2 +starch iodine-starch complex (blackish-blue) It is worthwhile to know that although vitamin C is very stable when dry, it is readily oxidized by air (oxygen) when in solution; therefore, a solution of vitamin C should not be exposed to air for long periods. The amount of vitamin C can be calculated by using the following conversion factor 41 mL of 12 (0.01 M) = 1.76 mg vitamin C OBJECTIVE To determine the amount of vitamin C that is present in certain commercial food products by the titration method. Pro to this laboratory, there are techniques that you should revieo. You uoll find the relevant techmiques in the following experiments: (a) Use of the Spectroline pipet filler is in Experiment 2; (b) buret use and titration techniques are im Experiment 19.
terps Tile logical antioxidant (reducing agent). It helps to keep the iron in the yme, prolyl hydroxylase, in the reduced form and, thereby, it helps to to prevent scurvy is ascorbic acid. It is a powerful Acave ingredient in fruits and intain th thesis of normal ions and broken blood vessels. e enzyme activity. Prolyl hydroxylase is essential for the collagen. In scurvy, the abnormal collagen causes skin Vitamin C cannot be synthesized tained from the diet ( in the human body and must be e.g., citrus fruits, broccoli, turnip greens, sweet ppers, tomatoes) or by taking synthetic vitamin C (e.g, vitamin C blets, "high-C" drinks, and other vitamin C-fortified commercial foods). he minimum recommended adult daily requirement of vitamin C to y is 60 mg. Some people, among them the late Linus Pauling, wice Nobel Laureate, suggested that very large daily doses (250-10,000 ng) of vitamin C could help prevent the common cold, or at least lessen he symptoms for many individuals. No reliable medical data support this At present, the human quantitative requirement for vitamin C is still revent scurv claim. In this experiment, the amount of vitamin C is determined quantita- tively by titrating the test solution with a water-soluble form of iodine, I5: controversial and requires further research. [I: 1:1: -(triioode ion) (1) :11:+:1:-- Expanded octets 569
Show transcribed image text 47 EXPERIMENT 47 Report Sheet 1. The kind of fruit drink appl Jwic 2. Molarity of iodine solution ad 3. Titration results Sample 1 (100 mL) Sample 2 (20.0 mL) Sample 3 (30.0 mL) a. Initial buret reading b. Final buret reading c. Total volume of iodine ,',ã¼, mL u. 2 mL 2.5 mL 4.amL 10.5 mL 10.5 mL 20.5 mL solution used: (b)-(a) 4. The weight of vitamin C in the fruit drink sample: [(3c) x 1.76 mg/mL] 5. Concentration of vitamin C in the fruit drink (mg/100 mL): [(4)/volume of drink] x 100 O,20,30 5S 6. Average concentration of vitamin Cin the fruit drink 52 mg/100 mL
Post-Lab Questions 1. Use the average concentration of vitamin C from your data, and calculate the volume that would satisfy your minimum daily requirement of vitamin C. Show your work 2. Vitamin C is ascorbie acid. If you examine the structure of ascorbic acid given in the Background t. In the structure shown, the ascorbic acid is section (equation 2), there is no -COOH grou - as a lactone or cyclic ester. Write the equation for the hydrolysis (cleavage by addition of water) of E ste the lactone to the carboxylic acid. 3. Write the net ionic equation for the conversion of iodine, la, to iodide, I. What kind of reaction is this? 4. Refer to the Background section and examine the chemical reaction for the oxidation of ascorbic acid (equation 2) a. What happened to the hydrogen atoms that were present in the ascorbic acid? b. Acid was added in the titration, but it is not present in equation (2) in a stoichiometric amount. So, what is the purpose of the acid? 5. The iodine for this experiment is in the form of 15, and not 12 (see equation (1), Background section]. Why is this form of iodine used? 577
ts for General Organic and Blochemistry Bettelheim/Landesberg Vitamin C is oxidized by 12 (as 15) according to the following chemical reaction: HO-CH- CH2OH HO-CH-CH,OH O. . o 2HI н он он Oxidized product (MW 174) (MW 254) Vitamin C (MW 176) As vitamin C is oxidized by iodine, I2 becomes reduced to I. When the end point is reached (no vitamin C is left), the excess of 12 will react with a starch indicator to form a starch-iodine complex, which is blackish-blue in color. I2 +starch iodine-starch complex (blackish-blue) It is worthwhile to know that although vitamin C is very stable when dry, it is readily oxidized by air (oxygen) when in solution; therefore, a solution of vitamin C should not be exposed to air for long periods. The amount of vitamin C can be calculated by using the following conversion factor 41 mL of 12 (0.01 M) = 1.76 mg vitamin C OBJECTIVE To determine the amount of vitamin C that is present in certain commercial food products by the titration method. Pro to this laboratory, there are techniques that you should revieo. You uoll find the relevant techmiques in the following experiments: (a) Use of the Spectroline pipet filler is in Experiment 2; (b) buret use and titration techniques are im Experiment 19.
terps Tile logical antioxidant (reducing agent). It helps to keep the iron in the yme, prolyl hydroxylase, in the reduced form and, thereby, it helps to to prevent scurvy is ascorbic acid. It is a powerful Acave ingredient in fruits and intain th thesis of normal ions and broken blood vessels. e enzyme activity. Prolyl hydroxylase is essential for the collagen. In scurvy, the abnormal collagen causes skin Vitamin C cannot be synthesized tained from the diet ( in the human body and must be e.g., citrus fruits, broccoli, turnip greens, sweet ppers, tomatoes) or by taking synthetic vitamin C (e.g, vitamin C blets, "high-C" drinks, and other vitamin C-fortified commercial foods). he minimum recommended adult daily requirement of vitamin C to y is 60 mg. Some people, among them the late Linus Pauling, wice Nobel Laureate, suggested that very large daily doses (250-10,000 ng) of vitamin C could help prevent the common cold, or at least lessen he symptoms for many individuals. No reliable medical data support this At present, the human quantitative requirement for vitamin C is still revent scurv claim. In this experiment, the amount of vitamin C is determined quantita- tively by titrating the test solution with a water-soluble form of iodine, I5: controversial and requires further research. [I: 1:1: -(triioode ion) (1) :11:+:1:-- Expanded octets 569