CHM361H5 Lecture Notes - Lecture 15: Equilibrium Constant, Enzyme
Document Summary
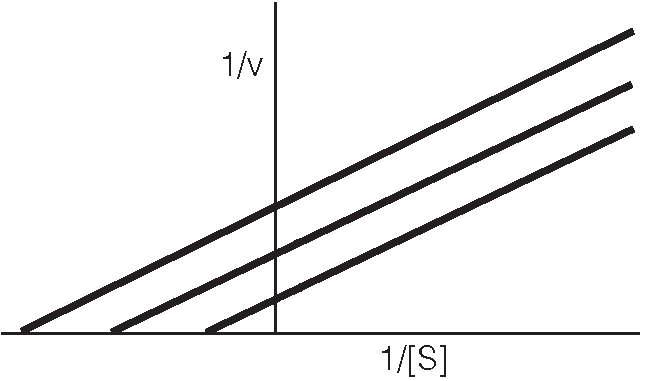
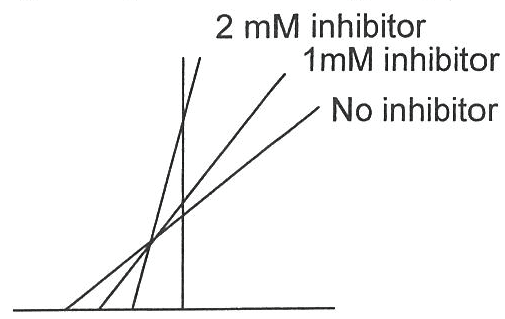
Vmax stays the same because you can flood the system with a higher concentration of substrate and throw off the enzyme. Inhibitors bind to both e and es. Or inhibitor could bind s in presence and absence of e, but this rare ! S and i binding occurs simultaneously to e at different sites. Because cannot throw off the enzyme by increasing the concentration of substrate. E + s es e + p. Ki dissociation constant for the inhibitor. This reaction decreases the concentration of es. Ki" dissociation constant for the inhibitor. No reaction of esi to form p. *** we will not talk about mixed non-competitive inhibition. Binding of s and i are independent. Affinity of e for s is unchanged. Effects of inhibitor i can not be overcome by high [s] Vmax decreases in presence of inhibitor i. When you add more inhibitor velocity decreases. If ki is bigger the velocity will increase.