CHMA11H3 Lecture Notes - Lecture 18: Boltzmann Constant, Isolated System, Closed System

36
CHMA11H3 Full Course Notes
Verified Note
36 documents
Document Summary
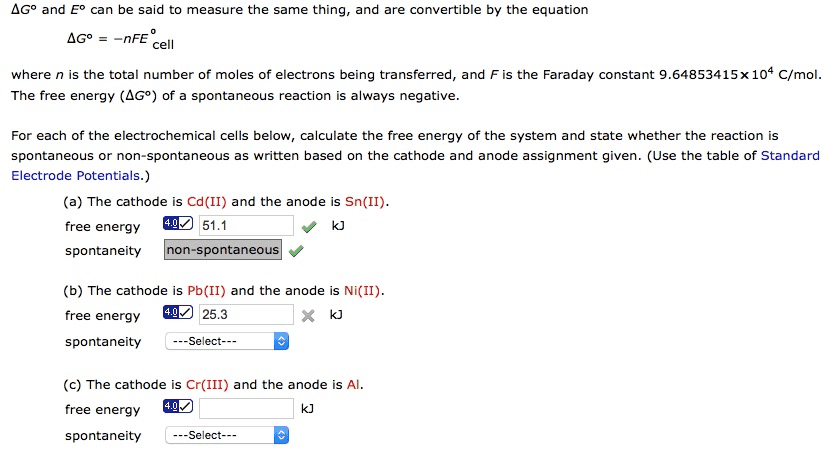
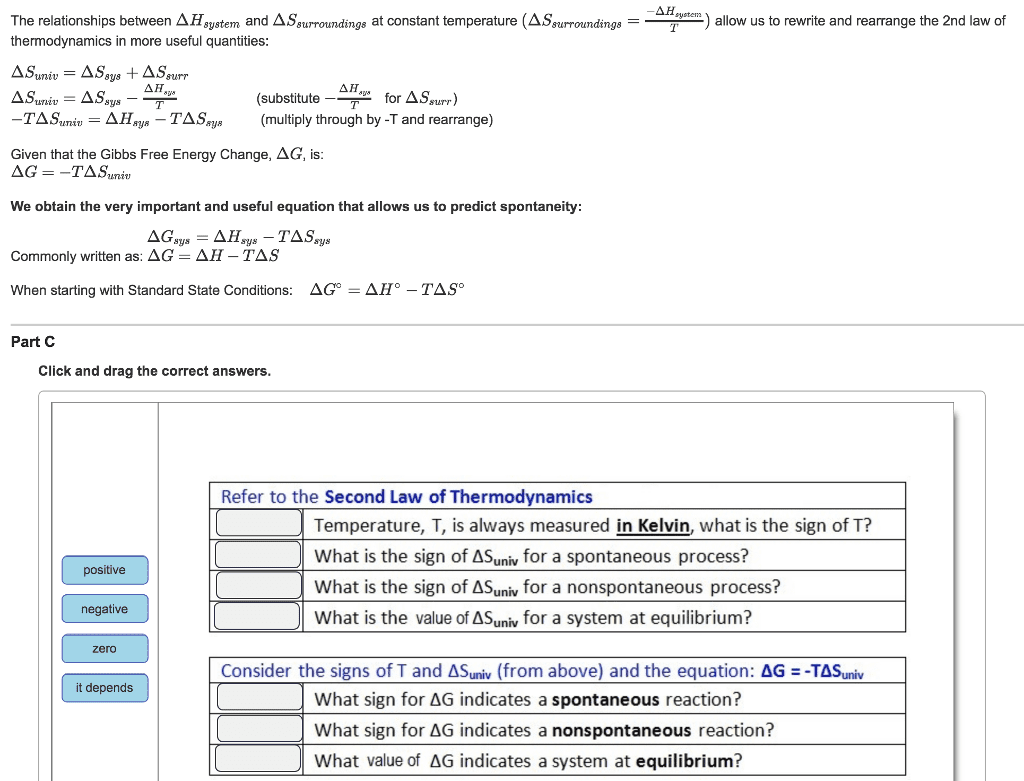
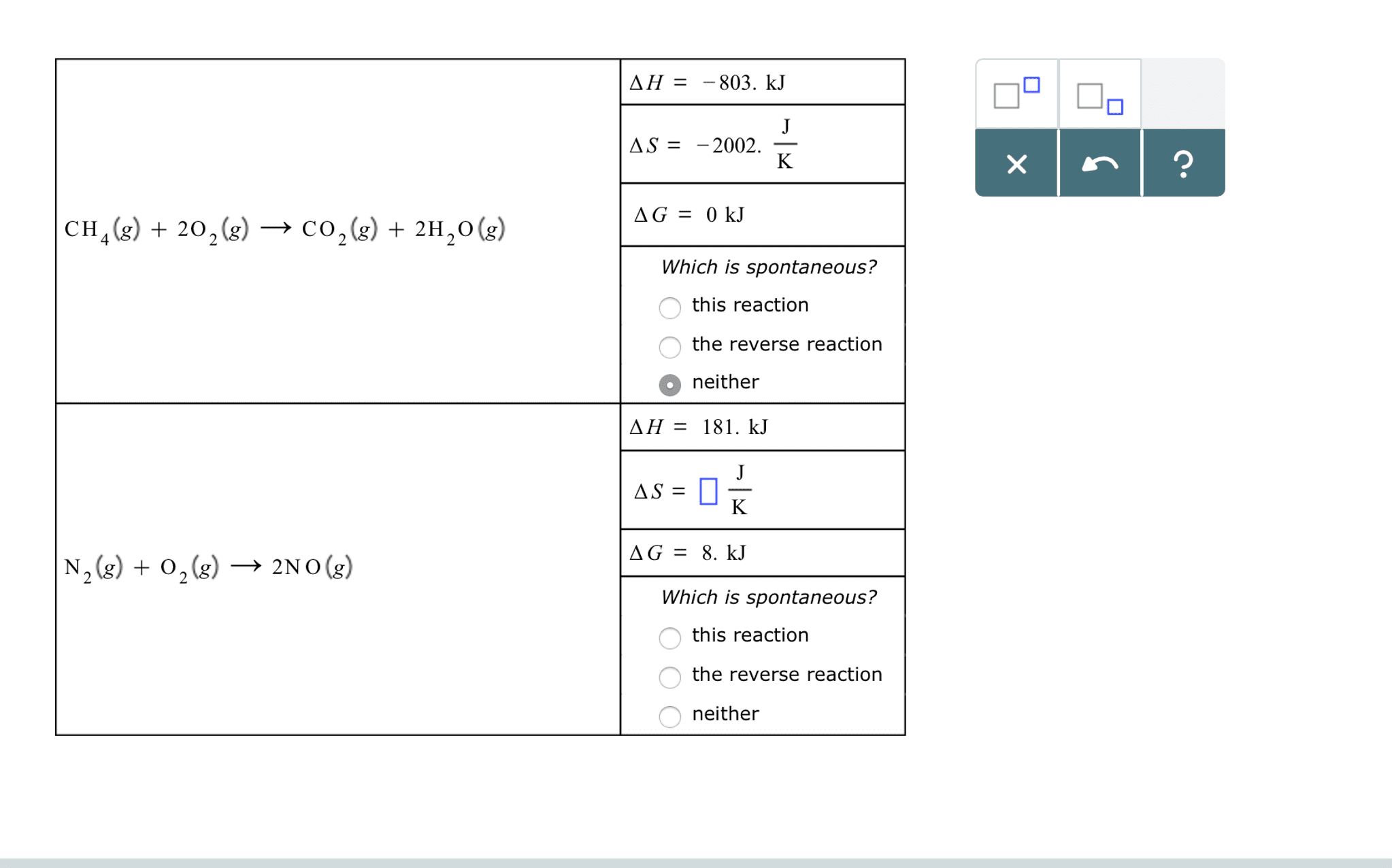
A spontaneous reaction is one that occurs by itself under a given set of conditions. A non-spontaneous reaction is one that requires some external action to be continuously applied in order for it to occur. Spontaneous changes will occur in the direction of the least potential energy. First law of thermodynamics: energy is neither created or destroyed, and the total energy in the universe is 0. Open system: allows both matter and energy to go in and out of the system. Closed system: allows only energy to enter and exit the system, not matter. Isolated system: allows neither matter nor energy to go in or out of the system. State function: a property which describes the state of a particle, and is path. Enthalpy is heat at constant pressure - review from chapter 6 independent ; no matter which way you measure it, it is always the same.