I just need to check my answers
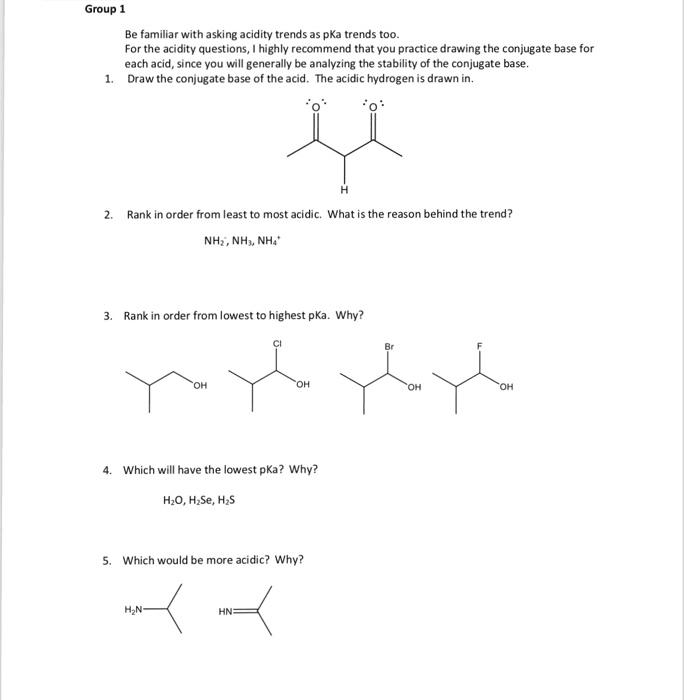
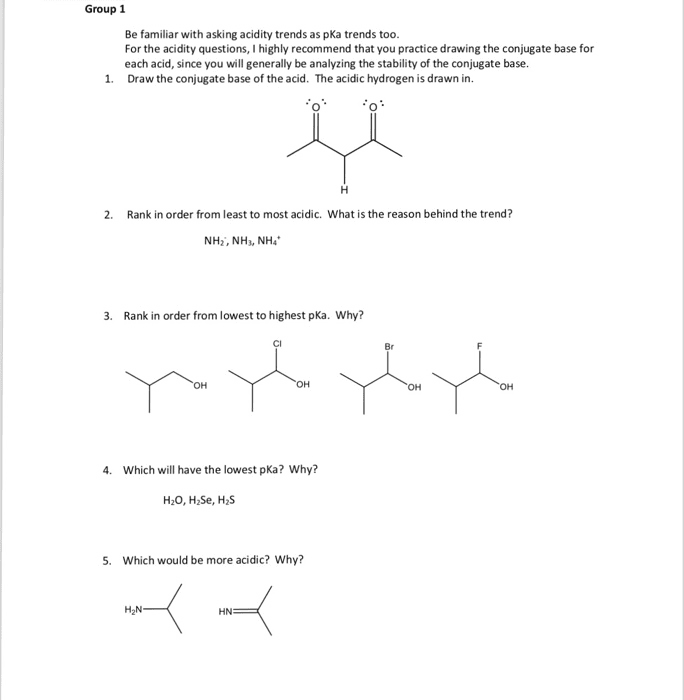
Group 1 Be familiar with asking acidity trends as pKa trends too. For the acidity questions, I highly recommend that you practice drawing the conjugate base for each acid, since you will generally be analyzing the stability of the conjugate base. Draw the conjugate base of the acid. The acidic hydrogen is drawn in. 1. 2. Rank in order from least to most acidic. What is the reason behind the trend? NH2, NH, NH4. 3. Rank in order from lowest to highest pKa. Why? Br он . Which will have the lowest pKa? Why? H20, H Se, H2S 5. Which would be more acidic? Why?
Show transcribed image text Group 1 Be familiar with asking acidity trends as pKa trends too. For the acidity questions, I highly recommend that you practice drawing the conjugate base for each acid, since you will generally be analyzing the stability of the conjugate base. Draw the conjugate base of the acid. The acidic hydrogen is drawn in. 1. 2. Rank in order from least to most acidic. What is the reason behind the trend? NH2, NH, NH4. 3. Rank in order from lowest to highest pKa. Why? Br он . Which will have the lowest pKa? Why? H20, H Se, H2S 5. Which would be more acidic? Why?