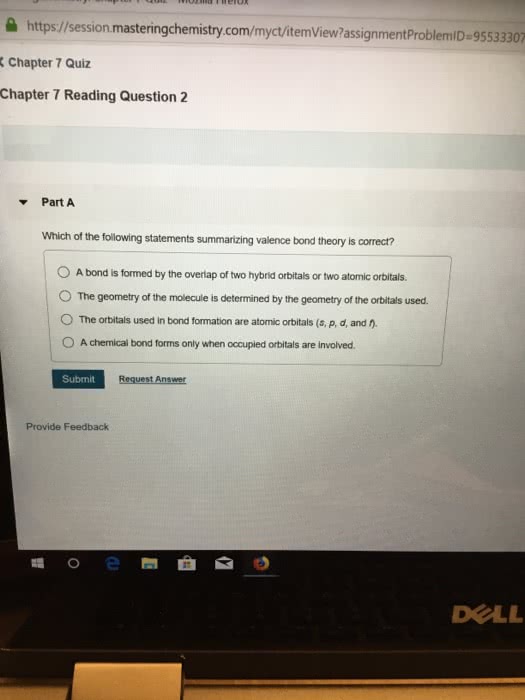
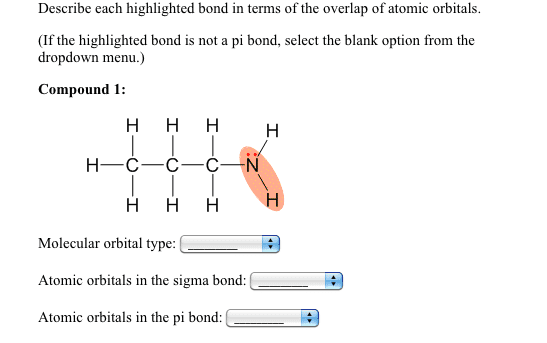
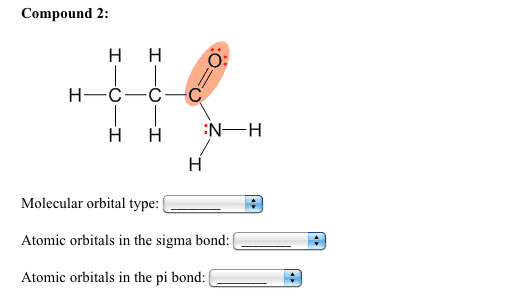
a) Does the [Fe(NH3)3(H2O)2 ] complex demonstrate geometric isomers? Does the [Fe(NH3)3(H2O)2 ] complex have an optical isomer?
Using the valence bond theory and hybrid orbitals show the orbital diagram of the complex. Use | to represent each electron of the Fe in hybrid and outer shell orbitals. Use : to represent each pair of electrons contributed by the NH3 and the H2O .
What type of hybrid orbitals must you use in the valence bond theory?
b) Does the [CoBr2I4] â3 complex demonstrate geometric isomers? Does the [CoBr2I4] â3 complex have an optical isomer?
Using the valence bond theory and hybrid orbitals show the orbital diagram of the complex. Use | to represent each electron of the Co in hybrid and outer shell orbitals. Use : to represent each pair of electrons contributed by the Br - and the I - .
What type of hybrid orbitals must you use in the valence bond theory?