CH110 Lecture Notes - Lecture 1: Silver Nitrate, Formula Unit, Aqueous Solution
Document Summary
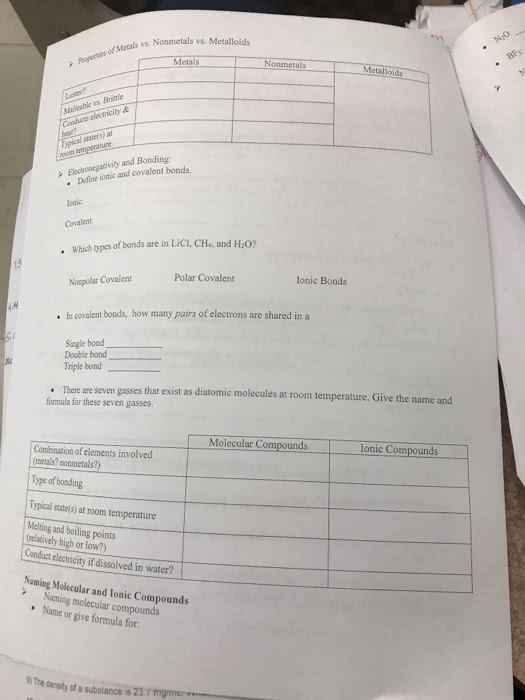
Chemistry: science that studies matter, its properties, and the changes it undergoes. Matter: anything that occupies space and has mass. Classification of matter: by physical state, by composition. Liquid: has definite volume but not shape, takes the shape of the container. Gas: has neither definite volume nor shape. Pure substances: pure substances (homogeneous, have the same composition throughout, can be elements or compounds. Elements: can"t be decomposed into simpler substance, composed of the same kind of atoms. Periodic table: all known elements are tabulated, each element is given a unique symbol. Symbols of elements: has 1 or 2 letters, first letter is always capitalized. Compounds: composed of 2 or more elements that are combined chemically (has to be a reaction, compounds can be decomposed into its elements. Mixtures: composed of 2 or more pure substances, components retain their properties (not chemically combined, can be homogeneous or heterogeneous. Homogenous mixtures: solutions are homogeneous mixtures of two or more pure substances.